- Home
- Telmitel 40
Telmitel 40
Telmitel 40 Telmisartan Tablets
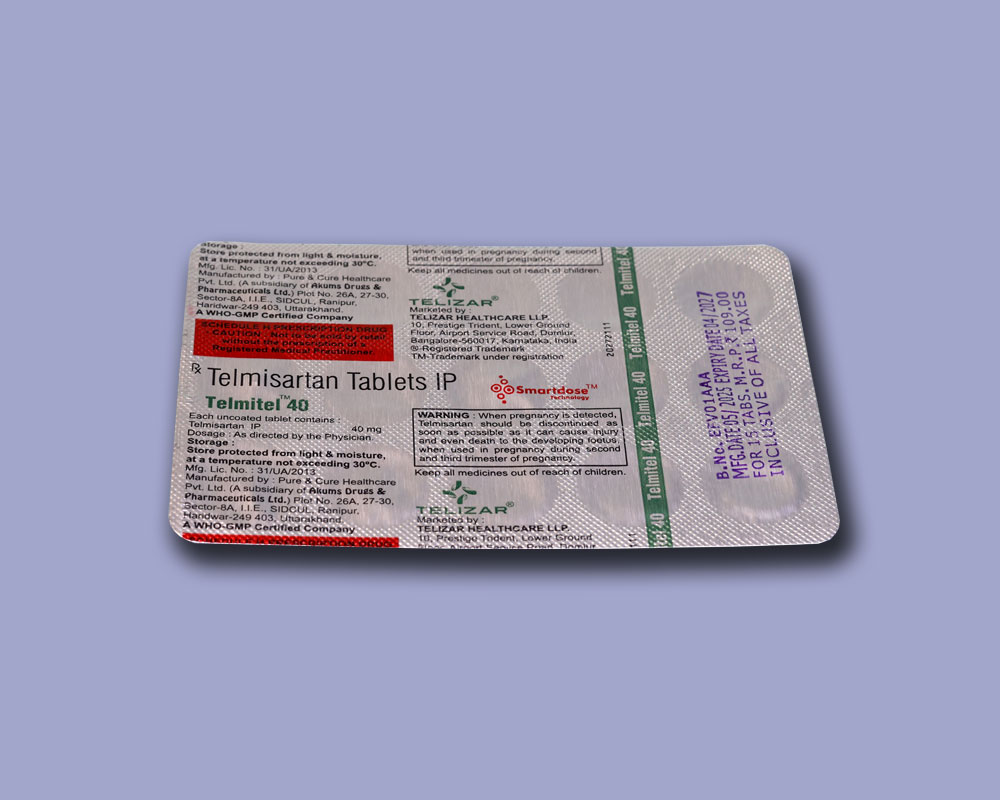
COMPOSITION
Each uncoated tablet contains:
Telmisartan IP
40 mg
PHARMACEUTICAL FORM
Each uncoated tablet contains.
THERAPEUTIC INDICATION
Indicated for the treatment of mild to moderate hypertension.
DOSAGE AND ADMINISTRATION
The recommended dose is 1 tablet once daily or as directed by the Physician.
Posology
The usually effective dose is 40 mg once daily. Some patients may already benefit at a daily dose of 20 mg. In cases where the target blood pressure is not achieved, the dose of telmisartan can be increased to a maximum of 80 mg once daily. Alternatively, telmisartan may be used in combination with thiazide-type diuretics such as hydrochlorothiazide, which has been shown to have an additive blood pressure lowering effect
with telmisartan. When considering raising the dose, it must be borne in mind that the maximum antihypertensive effect is generally attained four to eight weeks after the start of treatment.
Special populations:
Renal impairment
Limited experience is available in patients with severe renal impairment or haemodialysis. A lower starting dose of 20 mg is recommended in these patients. No posology adjustment is required for patients with mild to moderate renal impairment.
Hepatic impairment
Telmisartan is contraindicated in patients with severe hepatic impairment. In patients with mild to moderate hepatic impairment, the posology should not exceed 40 mg once daily.
Elderly
No dose adjustment is necessary for elderly patients.
Paediatric population
The safety and efficacy of Telmisartan in children and adolescents aged below 18 years have not been established.
Method of administration: For oral administration only.
Telmisartan tablets are for once-daily oral administration and should be taken with liquid, with or without food. Patients should try to take their daily dose at about the same time each day.
CONTRAINDICATIONS
Hypersensitivity to any of the active substance or to any of the excipients. Telmisartan is contraindicated in patients with:
- Second and third trimester of pregnancy.
- Biliary obstructive disorders.
- Severe hepatic impairment.
- The concomitant use of telmisartan with aliskiren-containing products is contraindicated in patients with diabetes mellitus or renal impairment (GFR<60 ml/min/1.73 m2).
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
Black Box Warning: Fetal Toxicity
When pregnancy is detected, discontinue telmisartan as soon as possible. Drugs that act directly on the renin-angiotensin system can cause injury and death to the developing fetus.
Pregnancy
Angiotensin II receptor antagonists should not be initiated during pregnancy. Unless continued angiotensin II receptor antagonist therapy is considered essential, patients planning pregnancy should be changed to alternative antihypertensive treatments which have an established safety profile for use in pregnancy. When pregnancy is diagnosed, treatment with angiotensin II receptor antagonists should be stopped immediately, and, if appropriate, alternative therapy should be started.
Hepatic impairment
Telmisartan is not to be given to patients with cholestasis, biliary obstructive disorders or severe hepatic impairment since telmisartan is mostly eliminated with the bile. These patients can be expected to have reduced hepatic clearance for telmisartan. Telmisartan should be used only with caution in patients with mild to moderate hepatic impairment.
Renovascular hypertension
There is an increased risk of severe hypotension and renal insufficiency when patients with bilateral renal artery stenosis or stenosis of the artery to a single functioning kidney are treated with medicinal products that affect the renin-angiotensin-aldosterone system.
Renal impairment and kidney transplantation
When Telmisartan is used in patients with impaired renal function, periodic monitoring of potassium and creatinine serum levels is recommended. There is no experience regarding the administration of Telmisartan in patients with recent kidney transplantation.
Intravascular hypovolaemia
Symptomatic hypotension, especially after the first dose of Telmisartan, may occur in patients who are volume and/or sodium depleted by vigorous diuretic therapy, dietary salt restriction, and diarrhoea or vomiting. Such conditions should be corrected before the administration of Telmisartan. Volume and/or sodium depletion should be corrected prior to administration of Telmisartan.
Dual blockade of the renin-angiotensin-aldosterone system (RAAS)
There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended. If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy.
Primary aldosteronism
Patients with primary aldosteronism generally will not respond to antihypertensive medicinal products acting through inhibition of the renin-angiotensin system. Therefore, the use of telmisartan is not recommended.
Aortic and mitral valve stenosis, obstructive hypertrophic cardiomyopathy
As with other vasodilators, special caution is indicated in patients suffering from aortic or mitral stenosis, or obstructive hypertrophic cardiomyopathy.
Diabetic patients treated with insulin or antidiabetics
In these patients’ hypoglycaemia may occur under telmisartan treatment. Therefore, in these patients an appropriate blood glucose monitoring should be considered; a dose adjustment of insulin or antidiabetics may be required, when indicated.
Hyperkalaemia
The use of medicinal products that affect the renin-angiotensin-aldosterone system may cause hyperkalaemia. In the elderly, in patients with renal insufficiency, in diabetic patients, in patients concomitantly treated with other medicinal products that may increase potassium levels, and/or in patients with intercurrent events, hyperkalaemia may be fatal.
Other
As with any antihypertensive agent, excessive reduction of blood pressure in patients with ischaemic cardiopathy or ischaemic cardiovascular disease could result in a myocardial infarction or stroke.
DRUG INTERACTION
Aliskiren
Do not co-administer aliskiren with telmisartan in patients with diabetes. Avoid use of aliskiren with telmisartan in patients with renal impairment (GFR <60 mL/min).
Digoxin
When telmisartan was co-administered with digoxin, median increases in digoxin peak plasma concentration (49%) and in trough concentration (20%) were observed. It is, therefore, recommended that digoxin levels be monitored when initiating, adjusting, and discontinuing telmisartan to avoid possible over- or under-digitalization. Lithium Reversible increases in serum lithium concentrations and toxicity have been reported during concomitant administration of lithium with angiotensin II receptor antagonists including telmisartan. Therefore, monitor serum lithium levels during concomitant use.
NSAIDs including Selective Cyclooxygenase-2 Inhibitors (COX-2 Inhibitors)
In patients who are elderly, volume-depleted (including those on diuretic therapy), or with compromised renal function, co-administration of non-steroidal anti-inflammatory drugs (NSAIDs), including selective COX-2 inhibitors, with ARBs, including telmisartan, may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Monitor renal function periodically receiving telmisartan and NSAID therapy. The antihypertensive effect of ARBs, including telmisartan may be attenuated by NSAIDs including selective COX-2 inhibitors.
Ramipril and Ramiprilat
Co-administration of telmisartan 80 mg once daily and ramipril 10 mg once daily to healthy subjects increases steady-state Cmax and AUC of ramipril 2.3- and 2.1-fold, respectively, and Cmax and AUC of ramiprilat 2.4-and 1.5-fold, respectively. In contrast, Cmax and AUC of telmisartan decrease by 31% and 16%, respectively. When co-administering telmisartan and ramipril, the response may be greater because of the possibly additive pharmacodynamic effects of the combined drugs, and also because of the increased exposure to ramipril and ramiprilat in the presence of telmisartan. Co-administration of telmisartan and ramipril is not recommended.
Potassium Sparing Diuretics or Potassium Supplements
ARBs such as telmisartan, attenuate diuretic induced potassium loss. Potassium sparing diuretics e.g. spironolactone, eplerenone, triamterene, or amiloride, potassium supplements, or potassium-containing salt substitutes may lead to a significant increase in serum potassium. If concomitant use is indicated because of documented hypokalemia they should be used with caution and with frequent monitoring of serum potassium.
Diuretics (Thiazide or Loop Diuretics) Prior treatment with high dose diuretics such as furosemide (loop diuretic) and hydrochlorothiazide (thiazide diuretic) may result in volume depletion and in a risk of hypotension when initiating therapy with telmisartan.
Other Antihypertensive Agents
The blood pressure lowering effect of telmisartan can be increased by concomitant use of other antihypertensive medicinal products.
Clinical trial data has shown that dual blockade of the RAS through the combined use of ACE-inhibitors, ARBs or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalemia and decreased renal function (including acute renal failure) compared to the use of a single RAS-acting agent.
Corticosteroids (Systemic Route)
Co-administered with telmisartan, can lead to reduction in the antihypertensive effect.
Other Drugs
Co-administration of telmisartan did not result in a clinically significant interaction with acetaminophen, amlodipine, glyburide, simvastatin, warfarin or ibuprofen. Telmisartan is not metabolized by the cytochrome P450 system and had no effects in vitro on cytochrome P450 enzymes, except for some inhibition of CYP2C19. Telmisartan is not expected to interact with drugs that inhibit cytochrome P450 enzymes; it is also not expected to interact with drugs metabolized by cytochrome P450 enzymes, except for possible inhibition of the metabolism of drugs metabolized by CYP2C19. Based on their pharmacological properties it can be expected that the following medicinal products may potentiate the hypotensive effects of all antihypertensives including telmisartan: Baclofen, amifostine. Furthermore, orthostatic hypotension may be aggravated by alcohol, barbiturates, narcotics or antidepressants.
FERTILITY, PREGNANCY AND LACTATION
Pregnancy
Teratogenic effects; Pregnancy categories C (first trimester) and D (second and third trimesters). The use of angiotensin II receptor antagonists is not recommended during the first trimester of pregnancy. The use of angiotensin II receptor antagonists is contraindicated during the second and third trimesters of pregnancy.
There are no adequate data from the use of Telmisartan Actavis in pregnant women. Studies in animals have shown reproductive toxicity. Epidemiological evidence regarding the risk of teratogenicity following exposure to ACE inhibitors during the first trimester of pregnancy has not been conclusive; however, a small increase in risk cannot be excluded. Whilst there is no controlled epidemiological data on the risk with angiotensin II receptor antagonists, similar risks may exist for this class of drugs. Unless continued angiotensin II receptor antagonist therapy is considered essential, patients planning pregnancy should be changed to alternative anti-hypertensive treatments which have an established safety profile for use in pregnancy. When pregnancy is diagnosed, treatment with angiotensin II receptor antagonists should be stopped immediately, and, if appropriate, alternative therapy should be started.
Exposure to angiotensin II receptor antagonist therapy during the second and third trimesters is known to induce human fetotoxicity (decreased renal function, oligohydramnios, skull ossification retardation) and neonatal toxicity (renal failure, hypotension, hyperkalaemia). Should exposure to angiotensin II receptor antagonists have occurred from the second trimester of pregnancy, ultrasound check of renal function and skull is recommended. Infants whose mothers have taken angiotensin II receptor antagonists should be closely observed for hypotension.
Breast-feeding
Because no information is available regarding the use of Telmisartan Actavis during breast-feeding, Telmisartan Actavis is not recommended and alternative treatments with better established safety profiles during breast-feeding are preferable, especially while nursing a newborn or preterm infant.
Fertility
In preclinical studies, no effects of telmisartan on male and female fertility were observed.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
When driving vehicles or operating machinery it should be taken into account that dizziness or drowsiness may occasionally occur when taking antihypertensive therapy such as Telmisartan.
UNDESIRABLE EFFECTS
The following adverse reaction is described elsewhere in labelling: Renal dysfunction upon use with ramipril.
Serious adverse drug reactions include anaphylactic reactions and angioedema which may occur rarely (> 1/10000 * to < 1/1000 ) and acute renal failure.
Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
Hypertension
Telmisartan has been evaluated for safety in more than 3700 patients, including 1900 treated for over 6 months and more than 1300 for over one year. Adverse experiences have generally been mild and transient in nature and have only infrequently required discontinuation of therapy. In placebo-controlled trials involving 1041 patients treated with various doses of telmisartan (20-160 mg) monotherapy for up to 12 weeks, an overall incidence of adverse events was similar to the patients treated with placebo. Adverse events occurring at an incidence of 1% in patients treated with telmisartan and at a greater rate than in patients treated with placebo, irrespective of their causal association, are: Back pain, sinusitis, upper respiratory tract infection, diarrhea, and pharyngitis.
In addition to the adverse events (mentioned above), the following events occurred at a rate of 1% but were at least as frequent in the placebo group: influenza-like symptoms, dyspepsia, myalgia, urinary tract infection, abdominal pain, headache, dizziness, pain, fatigue, coughing, hypertension, chest pain, nausea, and peripheral edema. Discontinuation of therapy because of adverse events was required in 2.8% of 1455 patients treated with telmisartan and 6.1% of 380 placebo patients in placebo-controlled clinical trials. The incidence of adverse events was not dose-related and did not correlate with gender, age, or race of patients. The incidence of cough occurring with telmisartan in six placebo-controlled trials was identical to that noted for placebo-treated patients (1.6%). In addition to those mentioned above, adverse events that occurred in more than 0.3% of 3500 patients treated with telmisartan monotherapy in controlled or open trials are listed below. It cannot be determined whether these events were causally related to telmisartan:
- Autonomic Nervous System: impotence, increased sweating, flushing.
- Body as a Whole: allergy, fever, leg pain, malaise.
- Cardiovascular: palpitation, dependent edema, angina pectoris, tachycardia, leg edema, abnormal ECG.
- CNS: insomnia, somnolence, migraine, vertigo, paresthesia, involuntary muscle contractions, hypoesthesia.
- Gastrointestinal: flatulence, constipation, gastritis, vomiting, dry mouth, hemorrhoids, gastroenteritis, enteritis, gastroesophageal reflux, toothache, nonspecific gastrointestinal disorders.
- Metabolic: gout, hypercholesterolemia, diabetes mellitus.
- Musculoskeletal: arthritis, arthralgia, leg cramps; Psychiatric: anxiety, depression, nervousness.
- Resistance Mechanism: infection, fungal infection, abscess, otitis media. Respiratory: asthma, bronchitis, rhinitis, dyspnea, epistaxis.
- Skin: dermatitis, rash, eczema, pruritus.
- Urinary: micturition frequency, cystitis.
- Vascular: cerebrovascular disorder.
- Special Senses: abnormal vision, conjunctivitis, tinnitus, earache.
Postmarketing experience
The following adverse reactions have been identified during post-approval use of telmisartan. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure. Decisions to include these reactions in labeling are typically based on one or more of the following factors: (1) seriousness of the reaction, (2) frequency of reporting, or (3) strength of causal connection to telmisartan. The most frequent spontaneously reported events include: headache, dizziness, asthenia, coughing, nausea, fatigue, weakness, edema, face edema, lower limb edema, angioneurotic edema, urticaria, hypersensitivity, sweating increased, erythema, chest pain, atrial fibrillation, congestive heart failure, myocardial infarction, blood pressure increased, hypertension aggravated, hypotension (including postural hypotension), hyperkalemia, syncope, dyspepsia, diarrhea, pain, urinary tract infection, erectile dysfunction, back pain, abdominal pain, muscle cramps (including leg cramps), myalgia, bradycardia, eosinophilia, thrombocytopenia, uric acid increased, abnormal hepatic function/liver disorder, renal impairment including acute renal failure, anemia, increased creatine phosphokinase (CPK), anaphylactic reaction, tendon pain (including tendonitis, tenosynovitis), drug eruption (toxic skin eruption mostly reported as toxicoderma, rash, and urticaria), hypoglycemia (in diabetic patients), and angioedema (with fatal outcome). Rare cases of rhabdomyolysis, interstitial lung disease and sepsis including fatal outcome have been reported in patients receiving ARBs, including telmisartan.
OVERDOSE
Limited data are available with regard to overdosage in humans. The most likely manifestations of overdosage with telmisartan tablets would be hypotension, dizziness, and tachycardia; bradycardia could occur from parasympathetic (vagal) stimulation. If symptomatic hypotension should occur, supportive treatment should be instituted. Telmisartan is not removed by hemodialysis.
PHARMACOLOGICAL PROPERTIES
Pharmacotherapeutic group: Angiotensin II Receptor Antagonists.
Telmisartan is a non-peptide angiotensin II receptor blocker (ARB). Angiotensin II is formed from angiotensin I in a reaction catalyzed by angiotensin-converting enzyme (ACE, kininase II). Angiotensin II is the principal pressor agent of the renin-angiotensin system (RAS), with effects that include vasoconstriction, stimulation of synthesis and release of aldosterone, cardiac stimulation, and renal reabsorption of sodium. Telmisartan blocks the vasoconstrictor and aldosterone-secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor in many tissues, such as vascular smooth muscle and the adrenal gland. Its action is therefore independent of the pathways for angiotensin II synthesis.
Mechanism of action
Telmisartan is an orally active and specific angiotensin II receptor (type AT1) antagonist. Telmisartan displaces angiotensin II with very high affinity from its binding site at the AT1 receptor subtype, which is responsible for the known actions of angiotensin II. Telmisartan does not exhibit any partial agonist activity at the AT1 receptor. Telmisartan selectively binds the AT1 receptor. The binding is long-lasting. Telmisartan does not show affinity for other receptors, including AT2 and other less characterized AT receptors. The functional role of these receptors is not known, nor is the effect of their possible overstimulation by angiotensin II, whose levels are increased by telmisartan. Plasma aldosterone levels are decreased by telmisartan. Telmisartan does not inhibit human plasma renin or block ion channels. Telmisartan does not inhibit angiotensin converting enzyme (kininase II), the enzyme which also degrades bradykinin. Therefore, it is not expected to potentiate bradykinin-mediated adverse events.
In human, an 80 mg dose of telmisartan almost completely inhibits the angiotensin II evoked blood pressure increase. The inhibitory effect is maintained over 24 hours and still measurable up to 48 hours.
PHARMACOKINETIC PROPERTIES
Absorption
Following oral administration, peak concentrations (Cmax) of telmisartan are reached in 0.5-1 hour after dosing. Food slightly reduces the bioavailability of telmisartan, with a reduction in the area under the plasma concentration time curve (AUC) of about 6% with the 40 mg tablet and about 20% after a 160 mg dose. The absolute bioavailability of telmisartan is dose-dependent. At 40 mg and 160 mg, the bioavailability was 42% and 58%, respectively. The pharmacokinetics of orally administered telmisartan are nonlinear over the dose range of 20-160 mg, with greater than proportional increases of plasma concentrations (Cmax and AUC) with increasing doses.
Distribution
Telmisartan is highly bound to plasma proteins (>99.5%), mainly albumin and (alpha)1-acid glycoprotein. Plasma protein binding is constant over the concentration range achieved with recommended doses. The volume of distribution for telmisartan is approximately 500 liters, indicating additional tissue binding.
Metabolism and Elimination
Following either intravenous or oral administration of 14C-labeled telmisartan, most of the administered dose (>97%) was eliminated unchanged in the feces via biliary excretion; only minute amounts were found in the urine (0.91% and 0.49% of total radioactivity, respectively).
Telmisartan is metabolized by conjugation to form a pharmacologically inactive acyl glucuronide; the glucuronide of the parent compound is the only metabolite that has been identified in human plasma and urine. After a single dose, the glucuronide represents approximately 11% of the measured radioactivity in plasma. The cytochrome P450 isoenzymes are not involved in the metabolism of telmisartan.
Total plasma clearance of telmisartan is >800 mL/min. Terminal half-life and total clearance appear to be independent of dose.
INCOMPATIBILITY
Not applicable.
PACKAGING INFORMATION
As per carton
STORAGE INSTRUCTIONS
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
Manufactured by:
Pure & Cure Healthcare Pvt. Ltd.
(A subsidiary of Akums Drugs & Pharmaceuticals Ltd.)
Plot No. 26A, 27-30, Sector-8A, I.I.E., SIDCUL, Ranipur, Haridwar-249 403, Uttarakhand.
A WHO-GMP Certified Company
Marketed by:
TELIZAR HEALTHCARE LLP.
10, Prestige Trident, Lower Ground Floor, Airport Service Road, Domlur, Bangalore-560017, Karnataka, India
Registered Trademark – TM-Trademark under registration
Other Products
Dipitel 5

Cilnidtel T 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.