- Home
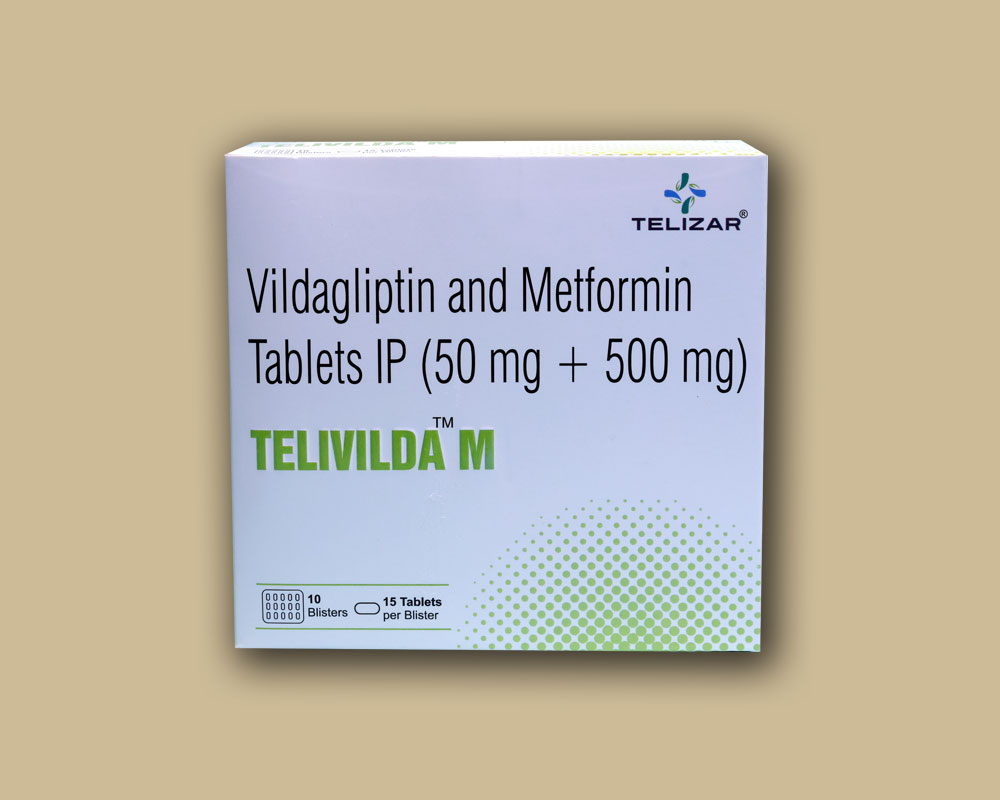
- Telivilda M
Telivilda M
Telivilda M Tablets
COMPOSITION
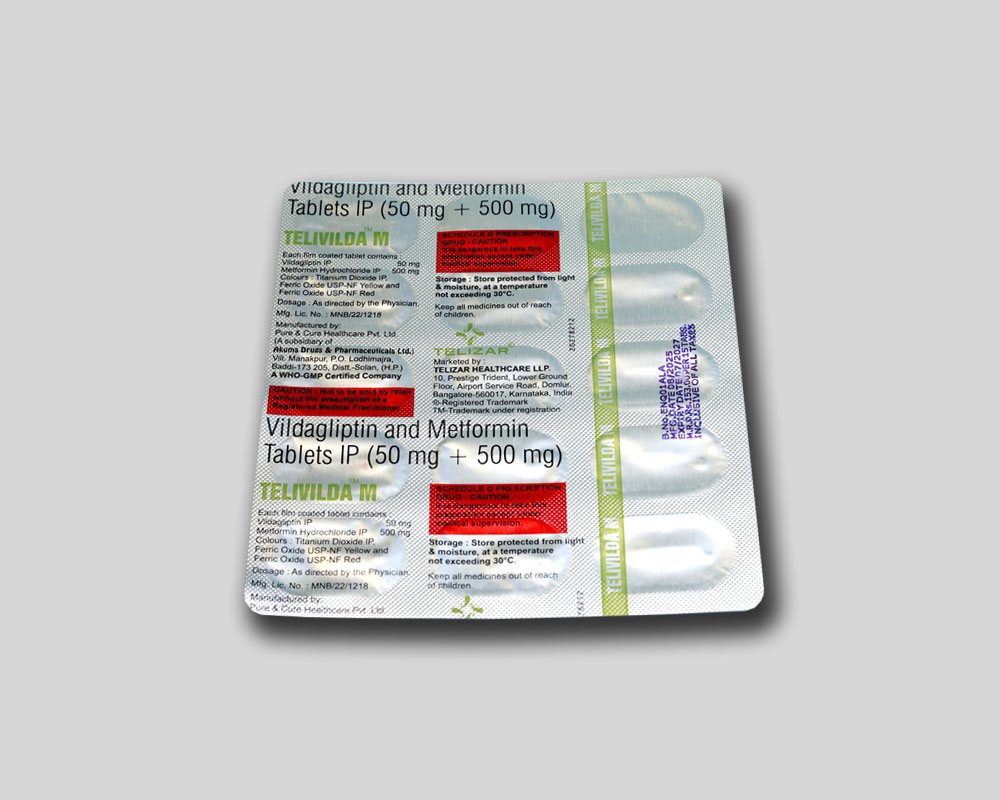
Each film coated tablet contains :
Vildagliptin IP 50 mg
Metformin Hydrochloride IP 500 mg
Colours : Titanium Dioxide IP, Ferric Oxide
USP-NF Yellow and Ferric Oxide USP-NF Red
DOSAGE FORM AND STRENGTH
Film Coated Tablet, Vildagliptin 50 mg & Metformin Hydrochloride 500 mg
THERAPEUTIC INDICATION
For the treatment of type-II diabetes mellitus when single drug therapy along with diet, exercise do not result in adequate glycemic control.
DOSAGE AND ADMINISTRATION
Posology
General
The use of antihyperglycemic therapy in the management of type 2 diabetes should be individualized on the basis of effectiveness and tolerability. When using Vildagliptin/Metformin do not exceed the maximum daily dose of vildagliptin (100 mg). The recommended starting dose of Vildagliptin/Metformin should be based on the patient’s condition and/or current regimen of vildagliptin and/or metformin hydrochloride.
Starting dose for patients inadequately controlled on vildagliptin monotherapy
Based on the usual starting doses of metformin hydrochloride (500 mg twice daily or 850 mg once daily), Vildagliptin/Metformin may be initiated at the 50 mg/500 mg tablet strength twice daily and gradually titrated after assessing the adequacy of therapeutic response.
Starting dose for patients inadequately controlled on metformin hydrochloride monotherapy
Based on the patient’s current dose of metformin hydrochloride, Vildagliptin/Metformin may be initiated at either the 50 mg/500 mg, 50 mg/850 mg or 50 mg/1,000 mg tablet strength twice daily. Starting dose for patients switching from combination therapy of vildagliptin plus metformin hydrochloride as separate tablets Vildagliptin/Metformin may be initiated with either the 50 mg/500 mg, 50 mg/850 mg or 50 mg/1,000 mg tablet strength based on the dose of vildagliptin or metformin already being taken. Special populations Elderly (≥ 65 years)
As metformin is excreted via the kidney, and elderly patients have a tendency to decreased renal function, elderly patients taking Vildagliptin/Metformin should have their renal function monitored regularly.
Patients with Renal Impairment
A GFR should be assessed before initiation of treatment with metformin-containing products (such as Vildagliptin/Metformin) and at least annually thereafter. In patients at increased risk of further progression of renal impairment and in the elderly, renal function should be assessed more frequently, e.g. every 3 to 6 months.
The maximum daily dose of metformin should preferably be divided into 2 to 3 daily doses. Factors that may increase the risk of lactic acidosis should be reviewed before considering initiation of metformin-containing products in patients with GFR<60 ml/min. vildagliptin +metformin is contraindicated in patients with GFR <30 ml/min because of its metformin component. The following dosing recommendations apply to metformin and vildagliptin, used separately or in combination, in patients with renal impairment. If no adequate strength of vildagliptin + metformin available, individual components should be used instead of the fixed dose combination.
Table 1: Dose adjustments in patients with renal impairment.
|
GFR ml/min |
Metformin |
Vildagliptin |
|
60-89 |
Maximum daily dose is 3000 mg*. Dose reduction may be considered if renal function declines. |
Maximum daily dose is 100 mg. |
|
45-59 |
Starting dose should not be more than 1000mg with a maximum daily dose of 2000 mg*. |
Maximum daily dose is 50 mg. |
|
30-44 |
Starting dose should not be more than 500mg with a maximum daily dose of 1000 mg. |
|
|
<30 |
Metformin is contraindicated. |
Hepatic impairment
Vildagliptin/Metformin should not be used in patients with hepatic impairment, including those with pre-treatment alanine aminotransferase (ALT) or aspartate aminotransferase (AST) > 3 times the upper limit of normal (ULN)
Paediatric population Vildagliptin/Metformin is not recommended for use in children and adolescents (< 18 years). The safety and efficacy of Vildagliptin/Metformin in children and adolescents (< 18 years) have not been established. No data are available.
Method of administration: For oral use only. Taking Vildagliptin/Metformin with or just after food may reduce gastrointestinal symptoms associated with metformin
CONTRAINDICATIONS
It is contraindicated in patients with known hypersensitivity to vildagliptin or metformin or to any other component of this formulation.
- Any type of acute metabolic acidosis (such as lactic acidosis, diabetic ketoacidosis)
- Diabetic pre-coma
- Severe renal failure (GFR < 30 ml/min)
- Acute conditions with the potential to alter renal function, such as:
- dehydration,
- severe infection,
- shock,
- Intravascular administration of iodinated contrast agents
- Acute or chronic disease which may cause tissue hypoxia, such as:
- cardiac or respiratory failure,
- recent myocardial infarction,
- Hepatic impairment
- Acute alcohol intoxication, alcoholism
- Breast-feeding
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
General
Vildagliptin & metformin is not a substitute for insulin in insulin-requiring patients and should not be used in patients with type 1 diabetes.
Lactic acidosis
Lactic acidosis, a very rare but serious metabolic complication, most often occurs at acute worsening of renal function, or cardiorespiratory illness or sepsis. Metformin accumulation occurs at acute worsening of renal function and increases the risk of lactic acidosis.In case of dehydration (severe diarrhoea or vomiting, fever or reduced fluid intake), metformin should be temporarily discontinued and contact with a health care professional is recommended. Medicinal products that can acutely impair renal function (such as antihypertensives, diuretics and NSAIDs) should be initiated with caution in metformin-treated patients. Other risk factors for lactic acidosis are excessive alcohol intake, hepatic insufficiency, inadequately controlled diabetes, ketosis, prolonged fasting and any conditions associated with hypoxia, as well as concomitant use of medicinal products that may cause lactic acidosis.
Patients and/or care-givers should be informed of the risk of lactic acidosis. Lactic acidosis is characterised by acidotic dyspnoea, abdominal pain, muscle cramps, asthenia and hypothermia followed by coma. In case of suspected symptoms, the patient should stop taking metformin and seek immediate medical attention. Diagnostic laboratory findings are decreased blood pH (< 7.35), increased plasma lactate levels (> 5 mmol/l) and an increased anion gap and lactate/pyruvate ratio.
Administration of iodinated contrast agents
Intravascular administration of iodinated contrast agents may lead to contrast-induced nephropathy, resulting in metformin accumulation and increased risk of lactic acidosis. Metformin should be discontinued prior to or at the time of the imaging procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Renal function GFR should be assessed before treatment initiation and regularly thereafter (see section 4.2). Metformin is contraindicated in patients with GFR < 30 ml/min and should be temporarily discontinued in the presence of conditions that alter renal function.
Concomitant medicinal products that may affect renal function, result in significant haemodynamic change, or inhibit renal transport and increase metformin systemic exposure, should be used with caution.
Hepatic impairment
Patients with hepatic impairment, including those with pre-treatment ALT or AST > 3x ULN, should not be treated with Vildagliptin/Metformin.
Liver enzyme monitoring Rare cases of hepatic dysfunction (including hepatitis) have been reported with vildagliptin. In these cases, the patients were generally asymptomatic without clinical sequelae and liver function tests (LFTs) returned to normal after discontinuation of treatment. LFTs should be performed prior to the initiation of treatment with Vildagliptin/Metformin in order to know the patient’s baseline value. Liver function should be monitored during treatment with Vildagliptin/Metformin at three-month intervals during the first year and periodically thereafter. Patients who develop increased transaminase levels should be monitored with a second liver function evaluation to confirm the finding and be followed thereafter with frequent LFTs until the abnormality(ies) return(s) to normal. Should an increase in AST or in ALT of 3x ULN or greater persist, withdrawal of Vildagliptin/Metformin therapy is recommended. Patients who develop jaundice or other signs suggestive of liver dysfunction should discontinue Vildagliptin/Metformin.
Following withdrawal of treatment with Vildagliptin/Metformin and LFT normalisation, treatment with Vildagliptin/Metformin should not be re-initiated.
Skin disorders
Skin lesions, including blistering and ulceration have been reported with vildagliptin in extremities of monkeys in non-clinical toxicology studies. Although skin lesions were not observed at an increased incidence in clinical trials, there was limited experience in patients with diabetic skin complications. Furthermore, there have been post-marketing reports of bullous and exfoliative skin lesions. Therefore, in keeping with routine care of the diabetic patient, monitoring for skin disorders, such as blistering or ulceration, is recommended.
Acute pancreatitis Use of vildagliptin has been associated with a risk of developing acute pancreatitis. Patients should be informed of the characteristic symptom of acute pancreatitis.
If pancreatitis is suspected, vildagliptin should be discontinued; if acute pancreatitis is confirmed, vildagliptin should not be restarted. Caution should be exercised in patients with a history of acute pancreatitis.
Hypoglycaemia Sulphonylureas are known to cause hypoglycaemia. Patients receiving vildagliptin in combination with a sulphonylurea may be at risk for hypoglycaemia. Therefore, a lower dose of sulphonylurea may be considered to reduce the risk of hypoglycaemia.
Surgery Metformin must be discontinued at the time of surgery under general, spinal or epidural anaesthesia. Therapy may be restarted no earlier than 48 hours following surgery or resumption of oral nutrition and provided that renal function has been re-evaluated and found to be stable.
DRUG INTERACTIONS
Vildagliptin
Vildagliptin has a low potential for interactions with co-administered medicinal products. Since vildagliptin is not a cytochrome P (CYP) 450 enzyme substrate and does not inhibit or induce CYP 450 enzymes, it is not likely to interact with active substances that are substrates, inhibitors or inducers of these enzymes.
Results from clinical trials conducted with the oral antidiabetics pioglitazone, metformin and glyburide in combination with vildagliptin have shown no clinically relevant pharmacokinetic interactions in the target population.
Drug-drug interaction studies with digoxin (P-glycoprotein substrate) and warfarin (CYP2C9 substrate) in healthy subjects have shown no clinically relevant pharmacokinetic interactions after co- administration with vildagliptin.
Drug-drug interaction studies in healthy subjects were conducted with amlodipine, ramipril, valsartan and simvastatin. In these studies, no clinically relevant pharmacokinetic interactions were observed after co-administration with vildagliptin. However, this has not been established in the target population.
Combination with ACE inhibitors
There may be an increased risk of angioedema in patients concomitantly taking ACE inhibitors.
As with other oral antidiabetic medicinal products the hypoglycaemic effect of vildagliptin may be reduced by certain active substances, including thiazides, corticosteroids, thyroid products and sympathomimetics.
Metformin
Combinations not recommended Alcohol
Alcohol intoxication is associated with an increased risk of lactic acidosis, particularly in cases of fasting, malnutrition or hepatic impairment.
Iodinated contrast agents
Metformin must be discontinued prior to or at the time of the imaging procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Combinations requiring precautions for use
Some medicinal products can adversely affect renal function which may increase the risk of lactic acidosis, e.g. NSAIDs, including selective cyclo-oxygenase (COX) II inhibitors, ACE inhibitors, angiotensin II receptor antagonists and diuretics, especially loop diuretics. When starting or using such products in combination with metformin, close monitoring of renal function is necessary.
Glucocorticoids, beta-2-agonists, and diuretics have intrinsic hyperglycaemic activity. The patient should be informed and more frequent blood glucose monitoring performed, especially at the beginning of treatment. If necessary, the dosage of Vildagliptin/Metformin may need to be adjusted during concomitant therapy and on its discontinuation.
Angiotensin converting enzyme (ACE) inhibitors may decrease the blood glucose levels. If necessary, the dosage of the antihyperglycaemic medicinal product should be adjusted during therapy with the other medicinal product and on its discontinuation.
Concomitant use of medicinal products that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g. organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors such as ranolazine, vandetanib, dolutegravir and cimetidine) could increase systemic exposure to metformin.
USE IN SPECIAL POPULATION
Pregnancy There are no adequate data from the use of Vildagliptin/Metformin in pregnant women. For vildagliptin studies in animals have shown reproductive toxicity at high doses. For metformin, studies in animals have not shown reproductive toxicity. Studies in animals performed with vildagliptin and metformin have not shown evidence of teratogenicity, but foetotoxic effects at maternotoxic doses. The potential risk for humans is unknown. Vildagliptin & metformin should not be used during pregnancy.
Breast-feeding Studies in animals have shown excretion of both metformin and vildagliptin in milk. It is unknown whether vildagliptin is excreted in human milk, but metformin is excreted in human milk in low amounts. Due to both the potential risk of neonate hypoglycaemia related to metformin and the lack of human data with vildagliptin, Vildagliptin/Metformin should not be used during breast-feeding.
Fertility No studies on the effect on human fertility have been conducted for Vildagliptin/Metformin.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
No studies on the effects on the ability to drive and use machines have been performed. Patients who may experience dizziness as an adverse reaction should avoid driving vehicles or using machines.
UNDESIRABLE EFFECTS
Dipeptidyl-peptidase-4 (DPP-4) inhibitors induced arthralgia: Dipeptidyl-peptidase-4 (DPP-4) inhibitors like sitagliptin, vildagliptin, Saxagliptin, etc induced arthralgia.
There have been no therapeutic clinical trials conducted with Vildagliptin/Metformin. However, bioequivalence of Vildagliptin/Metformin with co-administered vildagliptin and metformin has been demonstrated. The data presented here relate to the co-administration of vildagliptin and metformin, where vildagliptin has been added to metformin. There have been no studies of metformin added to vildagliptin.
Summary of the safety profile The majority of adverse reactions were mild and transient, not requiring treatment discontinuations. No association was found between adverse reactions and age, ethnicity, duration of exposure or daily dose.
Rare cases of hepatic dysfunction (including hepatitis) have been reported with vildagliptin. In these cases, the patients were generally asymptomatic without clinical sequelae and liver function returned to normal after discontinuation of treatment. In data from controlled monotherapy and add-on therapy trials of up to 24 weeks in duration, the incidence of ALT or AST elevations ≥ 3x ULN (classified as present on at least 2 consecutive measurements or at the final on-treatment visit) was 0.2%, 0.3% and 0.2% for vildagliptin 50 mg once daily, vildagliptin 50 mg twice daily and all comparators, respectively. These elevations in transaminases were generally asymptomatic, non-progressive in nature and not associated with cholestasis or jaundice.
Rare cases of angioedema have been reported on vildagliptin at a similar rate to controls. A greater proportion of cases were reported when vildagliptin was administered in combination with an ACE inhibitor. The majority of events were mild in severity and resolved with ongoing vildagliptin treatment.
Tabulated list of adverse reactions
Adverse reactions reported in patients who received vildagliptin in double-blind studies as monotherapy and add-on therapies are listed below by system organ class and absolute frequency. Adverse reactions listed in Table 5 are based on information available from the metformin Summary of Product Characteristics available in the EU. Frequencies are defined as very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1,000 to <1/100); rare (≥1/10,000 to <1/1,000); very rare (<1/10,000), not known (cannot be estimated from the available data). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness.
Table 1 Adverse reactions reported in patients who received vildagliptin 100 mg daily as add-on therapy to metformin compared to placebo plus metformin in double-blind studies (N=208)
|
Metabolism and nutrition disorders |
|
|
Common |
Hypoglycaemia |
|
Nervous system disorders |
|
|
Common |
Tremor |
|
Common |
Headache |
|
Common |
Dizziness |
|
Uncommon |
Fatigue |
|
Gastrointestinal disorders |
|
|
Common |
Nausea |
Description of selected adverse reactions
In controlled clinical trials with the combination of vildagliptin 100 mg daily plus metformin, no withdrawal due to adverse reactions was reported in either the vildagliptin 100 mg daily plus metformin or the placebo plus metformin treatment groups.
In clinical trials, the incidence of hypoglycaemia was common in patients receiving vildagliptin in combination with metformin (1%) and uncommon in patients receiving placebo + metformin (0.4%). No severe hypoglycaemic events were reported in the vildagliptin arms.
In clinical trials, weight did not change from baseline when vildagliptin 100 mg daily was added to metformin (+0.2 kg and -1.0 kg for vildagliptin and placebo, respectively). Clinical trials of up to more than 2 years’ duration did not show any additional safety signals or unforeseen risks when vildagliptin was added on to metformin.
Combination with a sulphonylurea
Table 2 Adverse reactions reported in patients who received vildagliptin 50 mg twice daily in combination with metformin and a sulphonylurea (N=157)
|
Metabolism and nutritional disorders |
|
|
Common |
Hypoglycaemia |
|
Nervous system disorders |
|
|
Common |
Dizziness, tremor |
|
Skin and subcutaneous tissue disorders |
|
|
Common |
Hyperhidrosis |
|
General disorders and administration site conditions |
|
|
Common |
Asthenia |
Description of selected adverse reactions
There were no withdrawals due to adverse reactions reported in the vildagliptin + metformin + glimepiride treatment group versus 0.6% in the placebo + metformin + glimepiride treatment group. The incidence of hypoglycaemia was common in both treatment groups (5.1% for the vildagliptin + metformin + glimepiride group versus 1.9% for the placebo + metformin + glimepiride group). One severe hypoglycaemic event was reported in the vildagliptin group.
At the end of the study, effect on mean body weight was neutral (+0.6 kg in the vildagliptin group and -0.1 kg in the placebo group).
Combination with insulin
Table 3 Adverse reactions reported in patients who received vildagliptin 100 mg daily in combination with insulin (with or without metformin) in double-blind studies (N=371)
|
Metabolism and nutrition disorders |
|
|
Common |
Decreased blood glucose |
|
Nervous system disorders |
|
|
Common |
Headache, chills |
|
Gastrointestinal disorders |
|
|
Common |
Nausea, gastro-oesophageal reflux disease |
|
Uncommon |
Diarrhoea, flatulence |
Description of selected adverse reactions
In controlled clinical trials using vildagliptin 50 mg twice daily in combination with insulin, with or without concomitant metformin, the overall incidence of withdrawals due to adverse reactions was 0.3% in the vildagliptin treatment group and there were no withdrawals in the placebo group.
The incidence of hypoglycaemia was similar in both treatment groups (14.0% in the vildagliptin group vs 16.4% in the placebo group). Two patients reported severe hypoglycaemic events in the vildagliptin group, and 6 patients in the placebo group.
At the end of the study, effect on mean body weight was neutral (+0.6 kg change from baseline in the vildagliptin group and no weight change in the placebo group).
Additional information on the individual active substances of the fixed combination
Vildagliptin
Table 4 Adverse reactions reported in patients who received vildagliptin 100 mg daily as monotherapy in double-blind studies (N=1855)
|
Infections and infestations |
|
|
Very rare |
Upper respiratory tract infection |
|
Very rare |
Nasopharyngitis |
|
Metabolism and nutrition disorders |
|
|
Uncommon |
Hypoglycaemia |
|
Nervous system disorders |
|
|
Common |
Dizziness |
|
Uncommon |
Headache |
|
Vascular disorders |
|
|
Uncommon |
Oedema peripheral |
|
Gastrointestinal disorders |
|
|
Uncommon |
Constipation |
|
Musculoskeletal and connective tissue disorders |
|
|
Uncommon |
Arthralgia |
Description of selected adverse reactions
The overall incidence of withdrawals from controlled monotherapy trials due to adverse reactions was no greater for patients treated with vildagliptin at doses of 100 mg daily (0.3%) than for placebo (0.6%) or comparators (0.5%).
In comparative controlled monotherapy studies, hypoglycaemia was uncommon, reported in 0.4% (7 of 1,855) of patients treated with vildagliptin 100 mg daily compared to 0.2% (2 of 1,082) of patients in the groups treated with an active comparator or placebo, with no serious or severe events reported.
In clinical trials, weight did not change from baseline when vildagliptin 100 mg daily was administered as monotherapy (-0.3 kg and -1.3 kg for vildagliptin and placebo, respectively). Clinical trials of up to 2 years’ duration did not show any additional safety signals or unforeseen risks with vildagliptin monotherapy.
Metformin
Table 5 Adverse reactions for metformin component
|
Metabolism and nutrition disorders |
|
|
Very rare |
Decrease of vitamin B₁₂ absorption and lactic acidosis* |
|
Nervous system disorders |
|
|
Common |
Metallic taste |
|
Gastrointestinal disorders |
|
|
Very common |
Nausea, vomiting, diarrhoea, abdominal pain and loss of appetite |
|
Hepatobiliary disorders |
|
|
Very rare |
Liver function test abnormalities or hepatitis** |
|
Skin and subcutaneous tissue disorders |
|
|
Very rare |
Skin reactions such as erythema, pruritus and urticaria |
|
*A decrease in vitamin B12 absorption with decrease in serum levels has been very rarely observed in patients treated long-term with metformin. Consideration of such aetiology is recommended if a patient present with megaloblastic anaemia. **Isolated cases of liver function test abnormalities or hepatitis resolving upon metformin discontinuation have baeen reported. |
|
Gastrointestinal adverse reactions occur most frequently during initiation of therapy and resolve spontaneously in most cases. To prevent them, it is recommended that metformin be taken in 2 daily doses during or after meals. A slow increase in the dose may also improve gastrointestinal tolerability.
Post-marketing experience
Table 6 Post-marketing adverse reactions
|
Gastrointestinal disorders |
|
|
Not known |
Pancreatitis |
|
Hepatobiliary disorders |
|
|
Not known |
Hepatitis (reversible upon discontinuation of the medicinal product) Abnormal liver function tests (reversible upon discontinuation of the medicinal product) |
|
Musculoskeletal and connective tissue disorders |
|
|
Not known |
Myalgia |
|
Skin and subcutaneous tissue disorders |
|
|
Not known |
Urticaria Exfoliative and bullous skin lesions, including bullous pemphigoid |
Reporting of side effects or suspected adverse reactions: If you experience any side effects, talk to your doctor or pharmacist or report to indiadrugsafety@akums.in. By reporting side effects, you can help provide more information on the safety of this product.
OVERDOSE
No data are available with regard to overdose of Vildagliptin/Metformin.
Vildagliptin
Information regarding overdose with vildagliptin is limited.
Symptoms
Information on the likely symptoms of overdose with vildagliptin was taken from a rising dose tolerability study in healthy subjects given vildagliptin for 10 days. At 400 mg, there were three cases of muscle pain, and individual cases of mild and transient paraesthesia, fever, oedema and a transient increase in lipase levels. At 600 mg, one subject experienced oedema of the feet and hands, and increases in creatine phosphokinase (CPK), AST, C-reactive protein (CRP) and myoglobin levels. Three other subjects experienced oedema of the feet, with paraesthesia in two cases. All symptoms and laboratory abnormalities resolved without treatment after discontinuation of the study medicinal product.
Metformin
A large overdose of metformin (or co-existing risk of lactic acidosis) may lead to lactic acidosis, which is a medical emergency and must be treated in hospital.
Management
The most effective method of removing metformin is haemodialysis. However, vildagliptin cannot be removed by haemodialysis, although the major hydrolysis metabolite (LAY 151) can. Supportive management is recommended.
PHARMACOLOGICAL PROPERTIES
Mechanism of Action
Vildagliptin, a member of the islet enhancer class, is a potent and selective dipeptidyl-peptidase-4 (DPP-4) inhibitor that improves glycemic control. Vildagliptin inhibition of DPP-4 results in increased fasting and postprandial endogenous levels of the incretin hormones GLP-1 (glucagon-like peptide 1) and GIP (glucose-dependent insulinotropic polypeptide).
Metformin hydrochloride decreases hepatic glucose production, decreases intestinal absorption of glucose and improves insulin sensitivity by increasing peripheral glucose uptake and utilization. Metformin hydrochloride stimulates intracellular glycogen synthesis by acting on glycogen synthase and increase the transport capacity of specific types of membrane glucose transporters (GLUT-1 and GLUT-4).
Pharmacodynamic properties
Vildagliptin
The administration of vildagliptin results in rapid and complete inhibition of DPP-4 activity. In patients with type 2 diabetes, administration of vildagliptin led to inhibition of DPP-4 enzyme activity for a 24-hour period.
By increasing the endogenous levels of these incretin hormones, vildagliptin enhances the sensitivity of beta cells to glucose, resulting in improved glucose-dependent insulin secretion. Treatment with 50 to 100 mg daily in patients with type 2 diabetes significantly improved markers of beta cell function. The degree of improvement in beta-cell function is dependent on the initial degree of impairment; in non-diabetic (normal glycemic) individuals, vildagliptin does not stimulate insulin secretion or reduce glucose levels.
By increasing endogenous GLP-1 levels, vildagliptin enhances the sensitivity of alpha cells to glucose, resulting in more glucose-appropriate glucagon secretion. The reduction in inappropriate glucagon during meals in turn attenuates insulin resistance.
The enhanced increase in the insulin/glucagon ratio during hyperglycemia due to increased incretin hormone levels results in a decrease in fasting and postprandial hepatic glucose production, leading to reduced glycemia.
The known effect of increased GLP-1 levels to delay gastric emptying is not observed with vildagliptin treatment. In addition, a reduction in postprandial lipemia that is not associated with vildagliptin’s incretin mediated effect to improve islet function has been observed.
Metformin Hydrochloride
Metformin hydrochloride improves glucose tolerance in patients with type 2 diabetes, lowering both basal and postprandial plasma glucose. Unlike sulfonylureas, metformin hydrochloride does not cause hypoglycemia in either patient with type 2 diabetes or normal subjects (except in special circumstances) and does not cause hyperinsulinemia.With metformin hydrochloride therapy, insulin secretion remains unchanged while fasting insulin levels and day-long plasma insulin response may actually decrease.
In humans, metformin hydrochloride has favorable effects on lipid metabolism, independent of its action on glycemia. This has been shown at therapeutic doses in controlled, medium-term or long-term clinical studies: metformin hydrochloride reduces total cholesterol, low-density lipoprotein cholesterol (LDLc) and triglyceride levels.
Pharmacokinetics properties
Absorption
Vildagliptin
Following oral administration in the fasting state, vildagliptin is rapidly absorbed with peak plasma concentrations observed at 1.7 hours. Food slightly delays the time to peak plasma concentration to 2.5 hours, but does not alter the overall exposure (AUC). Administration of vildagliptin with food resulted in a decreased Cmax (19%) compared to dosing in the fasting state. However, the magnitude of change is not clinically significant, so that vildagliptin can be given with or without food. The absolute bioavailability is 85%.
Metformin
After an oral dose of metformin, the maximum plasma concentration (Cmax) is achieved after about 2.5 h. Absolute bioavailability of a 500 mg metformin tablet is approximately 50-60% in healthy subjects. After an oral dose, the non-absorbed fraction recovered in faeces was 20-30%.
After oral administration, metformin absorption is saturable and incomplete. It is assumed that the pharmacokinetics of metformin absorption are non-linear. At the usual metformin doses and dosing schedules, steady state plasma concentrations are reached within 24-48 h and are generally less than 1 µg/ml. In controlled clinical trials, maximum metformin plasma levels (Cmax) did not exceed 4 µg/ml, even at maximum doses.
Food slightly delays and decreases the extent of the absorption of metformin. Following administration of a dose of 850 mg, the plasma peak concentration was 40% lower, AUC was decreased by 25% and time to peak plasma concentration was prolonged by 35 minutes. The clinical relevance of this decrease is unknown.
Distribution
Vildagliptin
The plasma protein binding of vildagliptin is low (9.3%) and vildagliptin distributes equally between plasma and red blood cells. The mean volume of distribution of vildagliptin at steady-state after intravenous administration (Vss) is 71 litres, suggesting extravascular distribution.
Metformin
Plasma protein binding is negligible. Metformin partitions into erythrocytes. The mean volume of distribution (Vd) ranged between 63-276 litres.
Biotransformation
Vildagliptin
Metabolism is the major elimination pathway for vildagliptin in humans, accounting for 69% of the dose. The major metabolite (LAY 151) is pharmacologically inactive and is the hydrolysis product of the cyano moiety, accounting for 57% of the dose, followed by the amide hydrolysis product (4% of dose). DPP-4 contributes partially to the hydrolysis of vildagliptin based on an in vivo study using DPP-4 deficient rats. Vildagliptin is not metabolised by CYP 450 enzymes to any quantifiable extent, and accordingly the metabolic clearance of vildagliptin is not anticipated to be affected by co-medications that are CYP 450 inhibitors and/or inducers. In vitro studies demonstrated that vildagliptin does not inhibit/induce CYP 450 enzymes. Therefore, vildagliptin is not likely to affect metabolic clearance of co- medications metabolised by CYP 1A2, CYP 2C8, CYP 2C9, CYP 2C19, CYP 2D6, CYP 2E1 or CYP 3A4/5.
Metformin
Metformin is excreted unchanged in the urine. No metabolites have been identified in humans.
Elimination
Vildagliptin
Following oral administration of [14C] vildagliptin, approximately 85% of the dose was excreted into the urine and 15% of the dose was recovered in the faeces. Renal excretion of the unchanged vildagliptin accounted for 23% of the dose after oral administration. After intravenous administration to healthy subjects, the total plasma and renal clearances of vildagliptin are 41 and 13 l/h, respectively. The mean elimination half-life after intravenous administration is approximately 2 hours. The elimination half-life after oral administration is approximately 3 hours.
Metformin
Metformin is eliminated by renal excretion. Renal clearance of metformin is > 400 ml/min, indicating that metformin is eliminated by glomerular filtration and tubular secretion. Following an oral dose, the apparent terminal elimination half-life is approximately 6.5 h. When renal function is impaired, renal clearance is decreased in proportion to that of creatinine and thus the elimination half-life is prolonged, leading to increased levels of metformin in plasma.
Linearity/non-linearity
The Cmax for vildagliptin and the area under the plasma concentrations versus time curves (AUC) increased in an approximately dose proportional manner over the therapeutic dose range.
Characteristics in patients
Gender: No clinically relevant differences in the pharmacokinetics of vildagliptin were observed between male and female healthy subjects within a wide range of age and body mass index (BMI). DPP-4 inhibition by vildagliptin is not affected by gender.
Age: In healthy elderly subjects (≥ 70 years), the overall exposure of vildagliptin (100 mg once daily) was increased by 32%, with an 18% increase in peak plasma concentration as compared to young healthy subjects (18-40 years). These changes are not considered to be clinically relevant, however. DPP-4 inhibition by vildagliptin is not affected by age.
Hepatic impairment: In subjects with mild, moderate or severe hepatic impairment (Child-Pugh A-C) there were no clinically significant changes (maximum ~30%) in exposure to vildagliptin.
Renal impairment: In subjects with mild, moderate, or severe renal impairment, systemic exposure to vildagliptin was increased (Cmax 8-66%; AUC 32-134%) and total body clearance was reduced compared to subjects with normal renal function.
Ethnic group: Limited data suggest that race does not have any major influence on vildagliptin pharmacokinetics.
INCOMPATIBILITIES
Not applicable.
STORAGE AND HANDLING INSTRUCTION
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
Other Products

Metfozar SR

Cilnidtel T 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.