- Home
- Dglifzar
Dglifzar
Dglifzar
1. GENERIC NAME
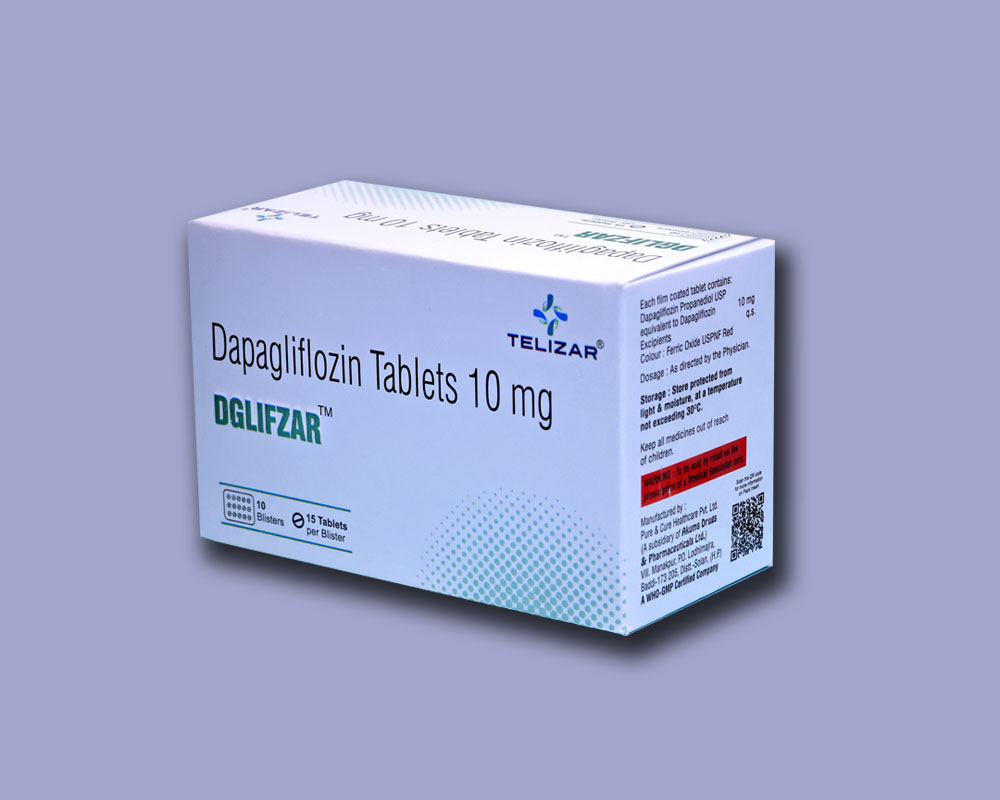
Dapagliflozin Tablets 10 mg
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Each film coated tablet contains:
Dapagliflozin Propanediol USP equivalent to Dapagliflozin 10 mg Excipients q.s.
Colour : Ferric Oxide USPNF Red
3. DOSAGE FORM AND STRENGTH
Film coated tablet
4. CLINICAL PARTICULARS
4.1THERAPEUTIC INDICATION
To reduce the risk of sustained eGFR and kidney disease, cardiovascular death, and hospitalization for heart failure in adult with chronic kidney disease at risk of progression” with condition that: – It is indicated in adults for the treatment of patients of chronic Kidney disease (CKD) upto eGFR of greater than or equal to 25 ml/min/1.73m2 .Below this, initiation of the treatment is not recommended, however the patients may continue 10 mg orally once daily to reduce the risk of eGFR decline, ESKD, CV death and hHF.
For the treatment of heart failure in adult.
4.2 Posology and method of administration Posology
The recommended dose is 1 tablet once daily or as directed by the Physician.
Method of administration: For oral use.
4.3 CONTRAINDICATIONS
Hypersensitivity to the active substance or to any of the excipients
4.4 SPECIAL WARNINGS AND PRECAUTIONS FOR USE
General
Dapagliflozin should not be used in patients with type 1 diabetes mellitus. Renal impairment
Due to limited experience, it is not recommended to initiate treatment with dapagliflozin in patients with GFR < 25 mL/min. The glucose lowering efficacy of dapagliflozin is dependent on renal function, and is reduced in patients with GFR < 45 mL/min and is likely absent in patients with severe renal impairment. In one study in patients with type 2 diabetes mellitus with moderate renal impairment (GFR < 60 mL/min), a higher proportion of patients treated with dapagliflozin had adverse reactions of increase in creatinine, phosphorus, parathyroid hormone (PTH) and hypotension, compared with placebo.
Hepatic impairment
There is limited experience in clinical studies in patients with hepatic impairment. Dapagliflozin exposure is increased in patients with severe hepatic impairment.
4.5 DRUG INTERACTION
Pharmacodynamic interactions
Diuretics
Dapagliflozin may add to the diuretic effect of thiazide and loop diuretics and may increase the risk of dehydration and hypotension.
Insulin and insulin secretagogues
Insulin and insulin secretagogues, such as sulphonylureas, cause hypoglycaemia. Therefore, a lower dose of insulin or an insulin secretagogue may be required to reduce the risk of hypoglycaemia when used in combination with dapagliflozin in patients with type 2 diabetes mellitus.
Pharmacokinetic interactions
The metabolism of dapagliflozin is primarily via glucuronide conjugation mediated by UDP glucuronosyltransferase 1A9 (UGT1A9). In in vitro studies, dapagliflozin neither inhibited cytochrome P450 (CYP) 1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP3A4, nor induced CYP1A2, CYP2B6 or CYP3A4. Therefore, dapagliflozin is not expected to alter the metabolic clearance of coadministered medicinal products that are metabolised by these enzymes.
Effect of other medicinal products on dapagliflozin
Interaction studies conducted in healthy subjects, using mainly a single-dose design, suggest that the pharmacokinetics of dapagliflozin are not altered by metformin, pioglitazone, sitagliptin, glimepiride, voglibose, hydrochlorothiazide, bumetanide, valsartan, or simvastatin. Following coadministration of dapagliflozin with rifampicin (an inducer of various active transporters and drug-metabolising enzymes) a 22% decrease in dapagliflozin systemic exposure (AUC) was observed, but with no clinically meaningful effect on 24-hour urinary glucose excretion. No dose adjustment is recommended. A clinically relevant effect with other inducers (e.g. carbamazepine, phenytoin, phenobarbital) is not expected. Following coadministration of dapagliflozin with Mefenamic acid (an inhibitor of UGT1A9), a 55% increase in dapagliflozin systemic exposure was seen, but with no clinically meaningful effect on 24-hour urinary glucose excretion. No dose adjustment is recommended.
Effect of dapagliflozin on other medicinal products
Dapagliflozin may increase renal lithium excretion and the blood lithium levels may be decreased. Serum concentration of lithium should be monitored more frequently after dapagliflozin initiation and dose changes. Please refer the patient to the lithium prescribing doctor in order to monitor serum concentration of lithium. In interaction studies conducted in healthy subjects, using mainly a single-dose design, dapagliflozin did not alter the pharmacokinetics of metformin, pioglitazone, sitagliptin, glimepiride, hydrochlorothiazide, bumetanide, valsartan, digoxin (a P-gp substrate) or warfarin (S-warfarin, a CYP2C9 substrate), or the anticoagulatory effects of warfarin as measured by INR. Combination of a single dose of dapagliflozin 20 mg and simvastatin (a CYP3A4 substrate) resulted in a 19% increase in AUC of simvastatin and 31% increase in AUC of simvastatin acid. The increase in simvastatin and simvastatin acid exposures are not considered clinically relevant.
Interference with 1,5-anhydroglucitol (1,5-AG) assay
Monitoring glycaemic control with 1,5-AG assay is not recommended as measurements of 1,5-AG are unreliable in assessing glycaemic control in patients taking SGLT2 inhibitors. Use of alternative methods to monitor glycaemic control is advised
4.6 FERTILITY, PREGNANCY AND LACTATION
Pregnancy There are no data from the use of dapagliflozin in pregnant women. Studies in rats have shown toxicity to the developing kidney in the time period corresponding to the second and third trimesters of human pregnancy. Therefore, the use of dapagliflozin is not recommended during the second and third trimesters of pregnancy. When pregnancy is detected, treatment with dapagliflozin should be discontinued.
Breast-feeding It is unknown whether dapagliflozin and/or its metabolites are excreted in human milk. Available pharmacodynamic/toxicological data in animals have shown excretion of dapagliflozin/metabolites in 7 milk, as well as pharmacologically-mediated effects in nursing. A risk to the newborns/infants cannot be excluded. Dapagliflozin should not be used while breast-feeding
Fertility The effect of dapagliflozin on fertility in humans has not been studied. In male and female rats, dapagliflozin showed no effects on fertility at any dose tested.
4.7 EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Dapagliflozin has no or negligible influence on the ability to drive and use machines. Patients should be alerted to the risk of hypoglycaemia when dapagliflozin is used in combination with a sulphonylurea or insulin.
4.8 UNDESIRABLE EFFECTS
Summary of the safety profile
Type 2 diabetes mellitus
In the clinical studies in type 2 diabetes, more than 15,000 patients have been treated with dapagliflozin. The primary assessment of safety and tolerability was conducted in a pre-specified pooled analysis of 13 short-term (up to 24 weeks) placebo-controlled studies with 2,360 subjects treated with dapagliflozin 10 mg and 2,295 treated with placebo. In the dapagliflozin cardiovascular outcomes study in type 2 diabetes mellitus, 8,574 patients received dapagliflozin 10 mg and 8,569 received placebo for a median exposure time of 48 months. In total, there were 30,623 patient-years of exposure to dapagliflozin. The most frequently reported adverse reactions across the clinical studies were genital infections.
Heart failure
In the dapagliflozin cardiovascular outcome study in patients with heart failure with reduced ejection fraction (DAPA-HF study), 2,368 patients were treated with dapagliflozin 10 mg and 2,368 patients with placebo for a median exposure time of 18 months. The patient population included patients with type 2 diabetes mellitus and without diabetes, and patients with eGFR ≥ 30 mL/min/1.73 m2. In the dapagliflozin cardiovascular outcome study in patients with heart failure with left ventricular ejection fraction > 40% (DELIVER), 3,126 patients were treated with dapagliflozin 10 mg and 3,127 patients with placebo for a median exposure time of 27 months. The patient population included patients with type 2 diabetes mellitus and without diabetes, and patients with eGFR ≥ 25 mL/min/1.73 m2. The overall safety profile of dapagliflozin in patients with heart failure was consistent with the known safety profile of dapagliflozin.
Chronic kidney disease
In the dapagliflozin renal outcome study in patients with chronic kidney disease (DAPA-CKD), 2,149 patients were treated with dapagliflozin 10 mg and 2,149 patients with placebo for a median exposure time of 27 months. The patient population included patients with type 2 diabetes mellitus and without diabetes, with eGFR ≥ 25 to ≤ 75 mL/min/1.73 m2, and albuminuria (urine albumin creatinine ratio [UACR] ≥ 200 and ≤ 5000 mg/g). Treatment was continued if eGFR fell to levels below 25 mL/min/1.73 m2. The overall safety profile of dapagliflozin in patients with chronic kidney disease was consistent with the known safety profile of dapagliflozin.
Table 1. Adverse reactions in placebo-controlled clinical studiesa and postmarketing experience
|
System organclass |
Very common |
Common* |
Uncommon** |
Rare |
Very rare |
|
Infections and infestations |
Vulvovaginitis, balanitis and related genital infections*,b,c Urinary tract infection*,b,d |
Fungal infection** |
Necrotising fasciitis of the perineum (Fournier’s gangrene)b,i |
||
|
Metabolism and nutrition disorders |
Hypoglycaemia (when used with SU or
insulin)b |
Volume depletionb,e Thirst** |
Diabetic ketoacidosis (when used in type 2 diabetes mellitus)b,i,k |
||
|
Nervous system disorders |
Dizziness |
||||
|
Gastrointestinal disorders |
Constipation** Dry mouth** |
||||
|
Skin and subcutaneous tissue disorders |
Rashj |
Angioedema |
|||
|
Musculoskeletal and connective tissue disorders |
Back pain* |
||||
|
Renal and urinary disorders |
Dysuria Polyuria*,f |
Nocturia** |
Tubulointerstitial nephritis |
||
|
Reproductive system and breast disorders |
Vulvovaginal pruritus** Pruritus genital** |
||||
|
Investigations |
Haematocrit increasedg Creatinine renal clearance decreased during initial treatmentb Dyslipidaemiah |
Blood creatinine increased during initial treatment**,b Blood urea increased** Weight decreased** |
a The table shows up to 24-week (short-term) data regardless of glycaemic rescue. b See corresponding subsection below for additional information. c Vulvovaginitis, balanitis and related genital infections includes, e.g. the predefined preferred terms: vulvovaginal mycotic infection, vaginal infection, balanitis, genital infection fungal, vulvovaginal candidiasis, vulvovaginitis, balanitis candida, genital candidiasis, genital infection, genital infection male, penile infection, vulvitis, vaginitis bacterial, vulval abscess
d Urinary tract infection includes the following preferred terms, listed in order of frequency reported: urinary tract infection, cystitis, Escherichia urinary tract infection, genitourinary tract infection, pyelonephritis, trigonitis, urethritis, kidney infection and prostatitis. e Volume depletion includes, e.g. the predefined preferred terms: dehydration, hypovolaemia, hypotension. f Polyuria includes the preferred terms: pollakiuria, polyuria, urine output increased. g Mean changes from baseline in haematocrit were 2.30% for dapagliflozin 10 mg versus –0.33% for placebo. Haematocrit values >55% were reported in 1.3% of the subjects treated with dapagliflozin 10 mg versus 0.4% of placebo subjects. h Mean percent change from baseline for dapagliflozin 10 mg versus placebo, respectively, was: total cholesterol 2.5% versus 0.0%; HDL cholesterol 6.0% versus 2.7%; LDL cholesterol 2.9% versus -1.0%; triglycerides -2.7% versus -0.7%.
j Adverse reaction was identified through postmarketing surveillance. Rash includes the following preferred terms, listed in order of frequency in clinical studies: rash, rash generalised, rash pruritic, rash macular, rash maculo-papular, rash pustular, rash vesicular, and rash erythematous. In active- and placebo-controlled clinical studies (dapagliflozin, N=5936, All control, N=3403), the frequency of rash was similar for dapagliflozin (1.4%) and all control (1.4%), respectively. k Reported in the cardiovascular outcomes study in patients with type 2 diabetes (DECLARE). Frequency is based on annual rate.
* Reported in ≥ 2% of subjects and ≥ 1% more and at least 3 more subjects treated with dapagliflozin 10 mg compared to placebo.
**Reported by the investigator as possibly related, probably related or related to study treatment and reported in ≥ 0.2% of subjects and ≥ 0.1% more and at least 3 more subjects treated with dapagliflozin 10 mg compared to placebo.
4.9 Overdose
Dapagliflozin did not show any toxicity in healthy subjects at single oral doses up to 500 mg (50 times the maximum recommended human dose). These subjects had detectable glucose in the urine for a dose-related period of time (at least 5 days for the 500 mg dose), with no reports of dehydration, hypotension or electrolyte imbalance, and with no clinically meaningful effect on QTc interval. The incidence of hypoglycaemia was similar to placebo. In clinical studies where once-daily doses of up to 100 mg (10 times the maximum recommended human dose) were administered for 2 weeks in healthy subjects and type 2 diabetes subjects, the incidence of hypoglycaemia was slightly higher than placebo and was not dose-related. Rates of adverse events including dehydration or hypotension were similar to placebo, and there were no clinically meaningful dose-related changes in laboratory parameters, including serum electrolytes and biomarkers of renal function. In the event of an overdose, appropriate supportive treatment should be initiated as dictated by the patient’s clinical status. The removal of dapagliflozin by haemodialysis has not been studied.
5. PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Drugs used in diabetes, sodium-glucose co-transporter 2 (SGLT2) inhibitors, ATC code: A10BK01 Mechanism of action Dapagliflozin is a highly potent (Ki: 0.55 nM), selective and reversible inhibitor of SGLT2. Inhibition of SGLT2 by dapagliflozin reduces reabsorption of glucose from the glomerular filtrate in the proximal renal tubule with a concomitant reduction in sodium reabsorption leading to urinary excretion of glucose and osmotic diuresis. Dapagliflozin therefore increases the delivery of sodium to the distal tubule which increases tubuloglomerular feedback and reduces intraglomerular pressure. This combined with osmotic diuresis leads to a reduction in volume overload, reduced blood pressure, and lower preload and afterload, which may have beneficial effects on cardiac remodelling and diastolic function, and preserve renal function. The cardiac and renal benefits of dapagliflozin are not solely dependent on the blood glucose-lowering effect and not limited to patients with diabetes as demonstrated in the DAPA-HF, DELIVER and DAPA-CKD studies. Other effects include an increase in haematocrit and reduction in body weight. Dapagliflozin improves both fasting and post-prandial plasma glucose levels by reducing renal glucose reabsorption leading to urinary glucose excretion. This glucose excretion (glucuretic effect) is observed after the first dose, is continuous over the 24-hour dosing interval and is sustained for the duration of treatment. The amount of glucose removed by the kidney through this mechanism is dependent upon the blood glucose concentration and GFR. Thus, in subjects with normal blood glucose, dapagliflozin has a low propensity to cause hypoglycaemia. Dapagliflozin does not impair normal endogenous glucose production in response to hypoglycaemia. Dapagliflozin acts independently of insulin secretion and insulin action. Improvement in homeostasis model assessment for beta cell function (HOMA beta-cell) has been observed in clinical studies with dapagliflozin. The SGLT2 is selectively expressed in the kidney. Dapagliflozin does not inhibit other glucose transporters important for glucose transport into peripheral tissues and is > 1,400 times more selective for SGLT2 versus SGLT1, the major transporter in the gut responsible for glucose absorption.
5.2 Pharmacokinetic properties
Absorption
Dapagliflozin was rapidly and well absorbed after oral administration. Maximum dapagliflozin plasma concentrations (Cmax) were usually attained within 2 hours after administration in the fasted state. Geometric mean steady-state dapagliflozin Cmax and AUCτ values following once daily 10 mg doses of dapagliflozin were 158 ng/mL and 628 ng h/mL, respectively. The absolute oral bioavailability of dapagliflozin following the administration of a 10 mg dose is 78%. Administration with a high-fat meal decreased dapagliflozin Cmax by up to 50% and prolonged Tmax by approximately 1 hour, but did not alter AUC as compared with the fasted state. These changes are not considered to be clinically meaningful. Hence, Dapagliflozin can be administered with or without food.
Distribution
Dapagliflozin is approximately 91% protein bound. Protein binding was not altered in various disease states (e.g. renal or hepatic impairment). The mean steady-state volume of distribution of dapagliflozin was 118 liters.
Biotransformation
Dapagliflozin is extensively metabolised, primarily to yield dapagliflozin 3-O-glucuronide, which is an inactive metabolite. Dapagliflozin 3-O-glucuronide or other metabolites do not contribute to the glucose-lowering effects. The formation of dapagliflozin 3-O-glucuronide is mediated by UGT1A9, an enzyme presents in the liver and kidney, and CYP-mediated metabolism was a minor clearance pathway in humans.
Elimination
The mean plasma terminal half-life (t1/2) for dapagliflozin was 12.9 hours following a single oral dose of dapagliflozin 10 mg to healthy subjects. The mean total systemic clearance of dapagliflozin administered intravenously was 207 mL/min. Dapagliflozin and related metabolites are primarily eliminated via urinary excretion with less than 2% as unchanged dapagliflozin. After administration of a 50 mg [14C]-dapagliflozin dose, 96% was recovered, 75% in urine and 21% in faeces. In faeces, approximately 15% of the dose was excreted as parent drug.
6. NON CLINICAL PROPERTIES
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, genotoxicity, carcinogenic potential and fertility. Dapagliflozin did not induce tumours in either mice or rats at any of the doses evaluated in two-year carcinogenicity studies.
7. DESCRIPTION
This product (tablet) contains: dapagliflozin as active ingredients and In adult aged 18 years and older with Type-II diabetic mellitus to improve glycemic control: As mono- therapy when diet and exercise alone do not provide adequate glycemic control in patients for whom use of metformin is considered inappropriate due to intolerance.
Dapagliflozin is a C-glycosyl comprising beta-D-glucose in which the anomeric hydroxy group is replaced by a 4-chloro-3-(4-ethoxybenzyl)phenyl group. Molecular Formula – C21H25ClO6
Molecular Weight – 408.9 g/mol
Chemical Structure
8. PHARMACEUTICAL PARTICULARS
8.1 Incompatibility
8.2 Shelf Life
Refer on carton
8.3 Packaging Information
Refer on carton
8.4 Storage instructions
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
9. PATIENT COUNSELLING INFORMATION
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you. Keep this leaflet. You may need to read it again.
If you have any further questions, ask your doctor, pharmacist or nurse.
This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours. If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet.
Other Products
Tezpara 650

Telipanta 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.