- Home
- Tenelizar
Tenelizar
Tenelizar 20 mg Tablet
COMPOSITION
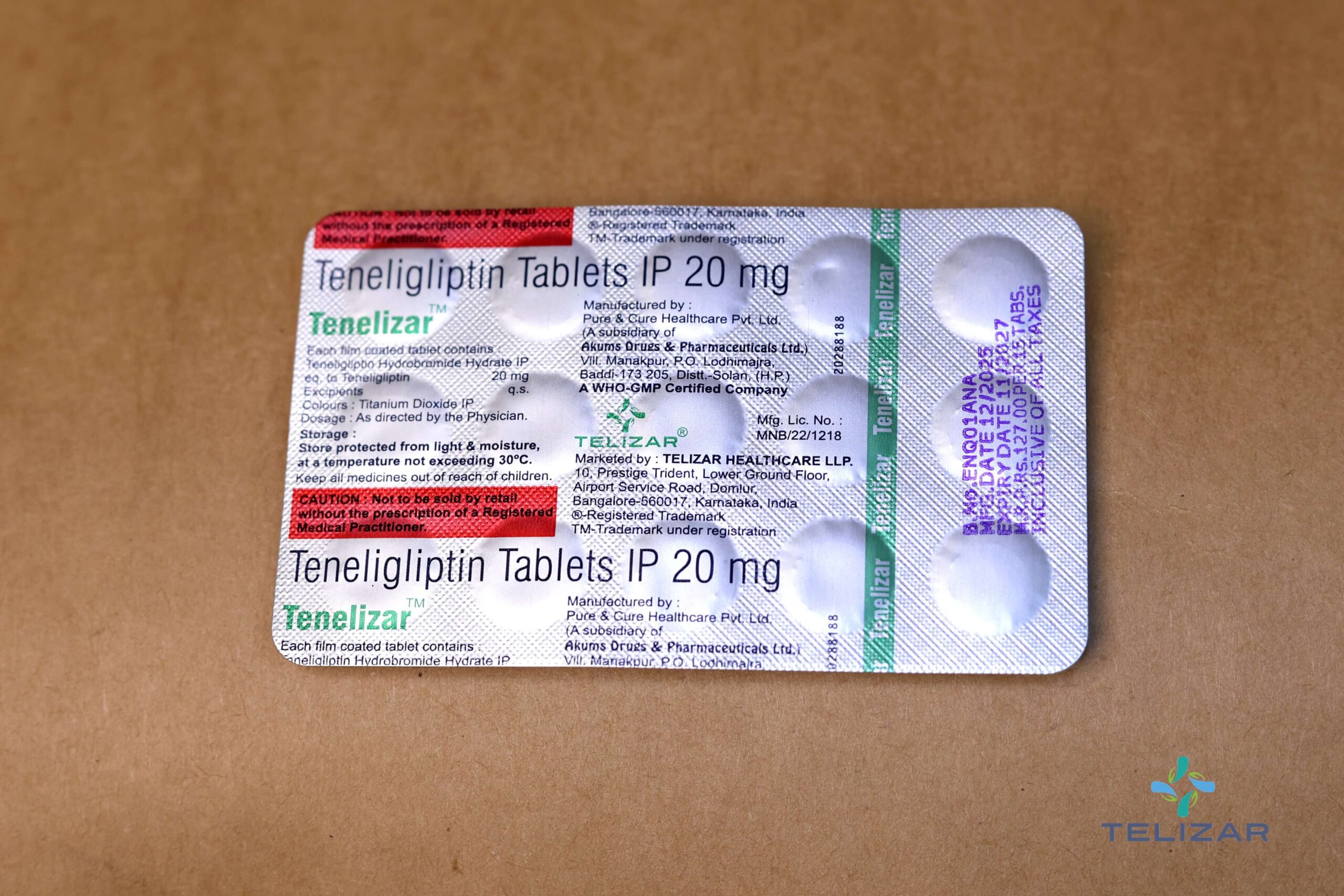
Each film coated tablet contains :
Teneligliptin Hydrobromide Hydrate IP
eq. to Teneligliptin 20 mg
Excipients q.s.
Colours : Titanium Dioxide IP
PHARMACEUTICAL FORM
film coated Tablet
THERAPEUTIC INDICATION
For the treatment of type 2 Diabetes Mellitus as a monotherapy adjunct to diet and Exercise.”
DOSE AND METHOD OF ADMINISTRATION
Posology
The usual adult dosage is 20 mg of teneligliptin administered orally once daily. If efficacy is insufficient, the dose may be increased up to 40 mg once daily while closely monitoring the clinical course.
Method of Administration: For oral administration only. Teneligliptin must be swallowed whole and never split, crushed or chewed
CONTRAINDICATIONS
Teneligliptin is contraindicated in the following: Any patient with a known hypersensitivity to teneligliptin or any of the components in the formulation, Severe ketosis, diabetic coma or history of diabetic coma, type 1 diabetic patients, Patients with severe infection, surgery, severe trauma (blood sugar control should preferably be done by insulin).
SPECIAL WARNINGS AND PRECAUTIONS
Teneligliptin should be administered carefully in the following: Patients with advanced liver failure (safety has not been established), Patients with congestive heart failure (NYHA category III-IV) (safety has not been established),
Patients with pituitary insufficiency or adrenal insufficiency, poor nutritional state, starvation, an irregular dietary intake, or debilitating condition, intense muscle movement or excessive alcohol intake (may cause low blood sugar), Patients with history of abdominal surgery or with a history of bowel obstruction (may cause bowel obstruction), Patients with arrhythmia, severe bradycardia or its history, patients with heart disease such as congestive heart failure or patients with low serum potassium, congenital prolonged QT syndrome, history of Torsades de pointes or patients using antiarrhythmic drugs (may cause QT prolongation), Patients using an insulin secretagogue (e.g., sulfonylurea) (risk of severe hypoglycaemia).
DRUG INTERACTIONS
Teneligliptin should be used with caution with drugs that can enhance the blood glucose lowering effect (like β blockers, MAO inhibitors, etc.) and attenuate the blood glucose lowering effect (like steroids, thyroid hormones, etc). On concomitant therapy with ketoconazole, the geometric least squares mean ratio (concomitant therapy/teneligliptin monotherapy) of Cmax and AUC0-t of unchanged plasma teneligliptin with their two-sided 90% CI is 1.37 [1.25, 1.50] and 1.49 [1.38, 1.60], respectively.
USE IN SPECIFIC POPULATION
Pregnancy
The safety of teneligliptin in pregnant women has not been established. Teneligliptin should be used in pregnant women or in women who may possibly be pregnant only if the expected therapeutic benefits outweigh the possible risks associated with treatment. (The safety of teneligliptin tablets in pregnant women has not been established. Furthermore, the transfer to embryo in animal studies (rats) has been reported).
Breast-feeding
Breast-feeding must be discontinued during administration of teneligliptin tablets in lactating women (transfer to milk in animal studies (rats) has been reported). Studies in lactating rats show that metformin is excreted into milk and reaches levels comparable to those in plasma.
Paediatric Use
Safety and effectiveness of teneligliptin in pediatric patients have not been established. The safety of teneligliptin in low birth weight baby, newborn baby, infant, or little child has not been established. (No usage experience). The safety and effectiveness in pediatric patients have not been established and Metformin is not recommended in pediatric patients below the age of 18 years. Thus, Teneligliptin/Metformin combination is not recommended in pediatric patients.
Geriatric Use
Start with lowest dose in elderly patients due to higher risks of lactic acidosis, poor renal and hepatic functions and therefore, teneligliptin should be administered carefully.
Patients with renal impairment
Teneligliptin/Metformin combination is not recommended in patients with renal impairment (e.g., serum creatinine levels greater than or equal to 1.5 mg/dL for men, greater than or equal to 1.4 mg/dL for women or abnormal creatinine clearance), which may also result from conditions such as cardiovascular collapse (shock), acute myocardial infarction, and septicemia.
The single administration of teneligliptin at 20 mg in patients with renal impairment revealed no remarkable changes in Cmax and t1/2 corresponding to the level of renal impairment. Compared with healthy adult subjects, the AUC0–∞ of subjects with mild renal impairment (50≤ creatinine clearance [Ccr] ≤ 80 mL/minute), moderate renal impairment (30 ≤ Ccr < 50 mL/minute), and severe renal impairment (Ccr < 30 mL/minute) was approximately 1.25 times, 1.68 times, and 1.49 times higher than that of healthy adult subjects, respectively. In addition, the AUC0–43h of patients with end-stage renal failure was approximately 1.16 times higher than that of healthy adult subjects. In addition, 15.6% of the total administration dose of teneligliptin was eliminated via hemodialysis.
A GFR should be assessed before initiation of treatment with metformin containing products and at least annually thereafter. In patients at an increased risk of further progression of renal impairment and in the elderly, renal function should be assessed more frequently, e.g. every 3-6 months.
Patients with hepatic impairment As determined from the pharmacokinetic characteristics of teneligliptin, the extent of increase in the exposure level of teneligliptin in patients with mild to moderate hepatic impairment will not pose any significant safety risk. Thus no dose adjustment is proposed in hepatic impaired patients. There was no clinical experience of teneligliptin in severe degree hepatic dysfunction patient. The presence of liver disease is a risk- factor for the development of lactic acidosis during metformin therapy, and the drug should be avoided in patients with hepatic insufficiency.
A single administration of teneligliptin 20 mg in patients with hepatic impairment revealed that the Cmax of subjects with mild hepatic impairment (Child-Pugh classification: total score 5–6) and moderate hepatic impairment (Child–Pugh classification: total score 7–9) was approximately 1.25 times and 1.38 times that of healthy adult subjects, respectively. Compared to healthy adult subjects, the AUC0–∞ of subjects with mild and moderate hepatic impairments was approximately 1.46 times and 1.59 times higher than that of healthy adult subjects, respectively. There have been no previous clinical studies using teneligliptin in patients with severe hepatic impairment (Child–Pugh classification: total score was greater than 9). Thus, specific caution is required when the drug is administered to patients with severe hepatic impairment.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Alertness and reactions may be impaired due to hypo- or hyperglycemia, especially when beginning or after altering treatment or when this tablet is not taken regularly. This may, for example, affect the ability to drive or to operate machinery.
UNDESIRABLE EFFECTS
Dipeptidyl-peptidase-4 (DPP-4) inhibitors induced arthralgia: Dipeptidyl-peptidase-4 (DPP-4) inhibitors like sitagliptin, vildagliptin, Teneligliptin, Saxagliptin, etc induced arthralgia.
The most common adverse reactions reported with teneligliptin are hypoglycemia and constipation. Other adverse reactions reported with teneligliptin are: Gastrointestinal Disorders: Intestinal obstruction, abdominal bloating, abdominal discomfort, nausea, abdominal pain, flatulence, stomatitis, gastric polyps, colon polyps, duodenal ulcer, reflux esophagitis, diarrhea, loss of appetite, increased amylase, lipase increased, acute pancreatitis.
Kidney and Urinary system: Proteinuria, urine ketone-positive. Skin and Subcutaneous Tissue Disorders: Eczema, rash, itching, allergic dermatitis. Investigations: Increase in AST, ALT, γ-GTP and ALP. Others: Increased CPK, increased serum potassium, fatigue, allergic rhinitis, elevation of serum uric acid
Reporting of side effects or suspected adverse reaction:
If you experience any side effects, talk to your doctor or pharmacist or report to indiadrugsafety@akums.in You can also report side effects directly via the National Pharmacovigilance Program of India by calling on 829 700 0060. By reporting side effects, you can help provide more information on the safety of this product.
OVERDOSE
In the event of an overdose, it is reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring (including obtaining an electrocardiogram), and institute supportive therapy as dictated by the patient’s clinical status.
PHARMACOLOGICAL PROPERTIES
Pharmacodynamic properties
Pharmacotherapeutic group: Drugs used in diabetes, blood glucose lowering drugs, excl. insulin’s.
Teneligliptin is a DPP-4 inhibitor, which is believed to exert its actions in patients with type 2 diabetes by slowing the inactivation of incretin hormones. Concentrations of the active intact hormones are increased by teneligliptin, thereby increasing and prolonging the action of these hormones. Incretin hormones, including glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP), are released by the intestine throughout the day, and levels are increased in response to a meal. These hormones are rapidly inactivated by the enzyme, DPP-4. The incretins are part of an endogenous system involved in the physiologic regulation of glucose homeostasis. When blood glucose concentrations are normal or elevated, GLP-1 and GIP increase insulin synthesis and release from pancreatic beta cells by intracellular signaling pathways involving cyclic AMP. GLP-1 also lowers glucagon secretion from pancreatic alpha cells, leading to reduced hepatic glucose production. By increasing and prolonging active incretin levels, teneligliptin increases insulin release and decreases glucagon levels in the circulation in a glucose-dependent manner.
Pharmacokinetic properties
Teneligliptin
After oral administration of a single 20 mg and 40 mg dose to healthy subjects, teneligliptin was rapidly absorbed, with peak plasma concentrations (mean T max) occurring at 1.8 hours and 1 hour postdose. Plasma AUC of teneligliptin increased in a dose-proportional manner. Following a single oral 20mg and 40mg dose to healthy volunteers, mean plasma AUC of teneligliptin was 2028.9 and 3705.1ng*hr/ml, Cmax was 187.2 and 382.4 ng/ml, and apparent terminal half-life (t1/2) was 24.2 and 20.8 hours. Plasma AUC of teneligliptin increased following 20mg doses at steady-state compared to the first dose. Coadministration with food reduces the Cmax by 20%, increases the Tmax from 1.1 to 2.6 hours but does not affect the AUC of teneligliptin as compared to that in the fasting state. The plasma protein binding rate is 77.6 –82.2%.
Following a 20mg single oral dose of [14C] teneligliptin, 45.4% of administered radioactivity was excreted in urine and 46.5% in faeces till 216
hours after dose. The cumulative urinary excretion rates for upto 120 hours for un-metabolized, M1, M2, and M3 were 14.8%, 17.7%, 1.4% and 1.9% respectively while the cumulative faecal excretion rates for un-metabolized, M1, M3, M4 and M5 were 26.1%, 4.0%, 1.6%, 0.3% and 1.3% respectively. CYP3A4, a cytochrome P450 isozyme and flavin-containing monooxygenases (FMO1 and FMO3) play major roles in the metabolism of teneligliptin. In vitro, teneligliptin exhibits a weak inhibitory effect for CYP2D6, CYP3A4, and FMO; however, it demonstrates no inhibitory effect for CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C8/9, CYP2C19, and CYP2E1. In addition, teneligliptin does not induce the expression of CYP1A2 or CYP3A4. About 34.4% of teneligliptin is excreted unchanged via the kidney and the remaining 65.6% teneligliptin is metabolized and eliminated via renal and hepatic excretion; 216 hours after the administration of 14C-labeled teneligliptin (20 mg), the cumulative excretion percentages of radioactive teneligliptin in urine and feces were 45.4% and 46.5%, respectively. Pharmacokinetic and pharmacodynamic properties of teneligliptin: The plasma concentrations of teneligliptin after the administration of teneligliptin at dosages of 10 or 20 mg once daily for 4 weeks revealed a median time to maximum concentration (Cmax) of 1.0 hour in both groups and a mean t1/2 of 20.8 and 18.9 hours, respectively. The maximum percentage of the inhibition in plasma DPP-4 activity was achieved within 2 hours after administration and was 81.3% and 89.7% in the 10 and 20 mg teneligliptin groups, respectively. The active GLP-1 concentration in the plasma in the 10 mg and 20 mg teneligliptin groups was higher than that in the placebo group throughout the day, even at 24 hours after administration. The AUC0–2h values for the active GLP-1 concentration after breakfast, lunch, and dinner were 8.0, 8.4, and 7.8 pmol h/L, respectively, in the 10 mg teneligliptin group,
and 8.3, 7.9, and 8.6 pmol h/L, respectively, in the 20 mg teneligliptin group. Thus, the increase in AUC0–2h for the active GLP- 1concentration after dinner was slightly greater in the 20 mg teneligliptin group than in the 10 mg teneligliptin group. Differences in the AUC0–2h for the active GLP-1 concentration between both the teneligliptin-treated groups and the placebo group were statistically significant.
INCOMPATIBILITIES
None stated.
STORAGE AND HANDLING INSTRUCTIONS
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
Other Products

Teliglim-MV

Telipanta 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.