- Home
- Sitaglipzar M
Sitaglipzar M
Sitaglipzar M
COMPOSITION
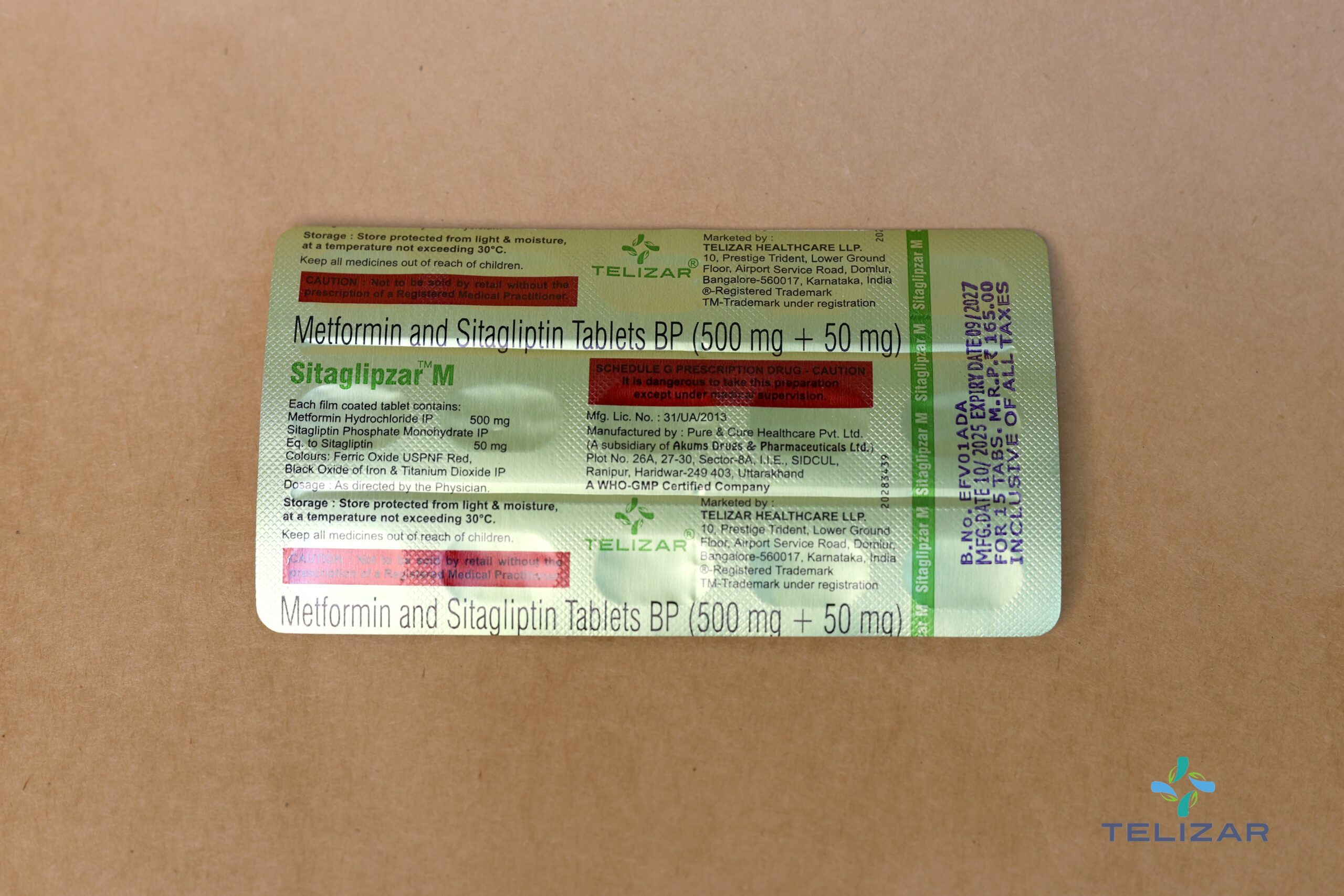
Each film coated tablet contains:
Metformin Hydrochloride IP 500 mg
Sitagliptin Phosphate Monohydrate IP
Eq. to Sitagliptin 50 mg
Colours: Ferric Oxide USPNF Red, Black Oxide of Iron & Titanium Dioxide IP
PHARMACEUTICAL FORM
Film Coated Tablet
THERAPEUTIC INDICATION
As an adjunct to diet & exercise to improve glycemic control in patients with type-2 diabetes mellitus.
- FDC is indicated as triple combination therapy with a PPARy agonist (i.e a thiazolidinedione) as an adjunct to diet and exercise in patients inadequtely controlled on their maximal tolerated dose of metfomin and a PPARy
- FDC is also indicated as add-on to insulin (i.e triple combination therapy) as an adjunct to diet and exercise to improve glycemic control in patients when stable dosage of insulin and metformin alone do not provide adequte
DOSAGE AND METHOD OF ADMINISTRATION
Recommended Dosing
Take Sitagliptin Phosphate & Metformin Hydrochloride Tablets orally twice daily with meals.
Individualize the dosage of Sitagliptin Phosphate & Metformin Hydrochloride Tablets on the basis of the patient’s current regimen, effectiveness, and tolerability. The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin hydrochloride (Hcl).
Do not split or divide Sitagliptin Phosphate & Metformin Hydrochloride Tablets. The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin.
The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of Sitagliptin Phosphate & Metformin Hydrochloride Tablets is 50 mg sitagliptin and 1000 mg metformin HCl twice daily.
Recommendations for Use in Renal Impairment
Assess renal function prior to initiation of Sitagliptin Phosphate & Metformin Hydrochloride Tablets and periodically thereafter. Sitagliptin Phosphate & Metformin Hydrochloride Tablets is contraindicated in patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m2.
Sitagliptin Phosphate & Metformin Hydrochloride Tablets is not recommended in patients with an eGFR between 30 and less than 45 mL/min/1.73 m2 because these patients require a lower dosage of sitagliptin than what is available in the fixed combination Sitagliptin Phosphate & Metformin Hydrochloride Tablets product.
Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue Sitagliptin Phosphate & Metformin Hydrochloride Tablets at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2 ; in patients with a history of liver disease, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Reevaluate eGFR 48 hours after the imaging procedure; restart Sitagliptin Phosphate & Metformin Hydrochloride Tablets if renal function is stable.
Method of administration: For Oral administration only.
CONTRAINDICATIONS
Sitagliptin & Metformin Tablets is contraindicated in patients with:
- hypersensitivity to the active substances or to any of the excipients – any type of acute metabolic acidosis (such as lactic acidosis, diabetic ketoacidosis);
- diabetic pre-coma;
- severe renal failure (GFR< 30 mL/min)
- acute conditions with the potential to alter renal function such as:
- dehydration,
- severe infection,
- shock,
- intravascular administration of iodinated contrast agents- acute or chronic disease which may cause tissue hypoxia such as:
- cardiac or respiratory failure,
- recent myocardial infarction,
- shock;- hepatic impairment;
- acute alcohol intoxication, alcoholism;
- breast-feeding.
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
General
Sitagliptin & Metformin Tablets should not be used in patients with type 1 diabetes and must not be used for the treatment of diabetic ketoacidosis.
Acute pancreatitis
Use of DPP-4 inhibitors has been associated with a risk of developing acute pancreatitis. Patients should be informed of the characteristic symptom of acute pancreatitis: persistent, severe abdominal pain. Resolution of pancreatitis has been observed after discontinuation of sitagliptin (with or without supportive treatment), but very rare cases of necrotising or haemorrhagic pancreatitis and/or death have been reported. If pancreatitis is suspected, Sitagliptin Phosphate & Metformin Hydrochloride Tablets and other potentially suspect medicinal products should be discontinued; if acute pancreatitis is confirmed, Sitagliptin Phosphate & Metformin Hydrochloride Tablets should not be restarted. Caution should be exercised in patients with a history of pancreatitis.
Lactic acidosis
Lactic acidosis, a rare but serious metabolic complication, most often occurs at acute worsening of renal function or cardiorespiratory illness or sepsis. Metformin accumulation occurs at acute worsening of renal function and increases the risk of lactic acidosis.
In case of dehydration (severe vomiting, diarrhoea, fever or reduced fluid intake), metformin should be temporarily discontinued and contact with a health care professional is recommended.
Medicinal products that can acutely impair renal function (such as antihypertensives, diuretics and NSAIDs) should be initiated with caution in metformin-treated patients. Other risk factors for lactic acidosis are excessive alcohol intake, hepatic insufficiency, inadequately controlled diabetes, ketosis, prolonged fasting and any conditions associated with hypoxia, as well as concomitant use of medicinal products that may cause lactic acidosis. Patients and/or care-givers should be informed of the risk of lactic acidosis. Lactic acidosis is characterised by acidotic dyspnoea, abdominal pain, muscle cramps, asthenia and hypothermia followed by coma. In case of suspected symptoms, the patient should stop taking metformin and seek immediate medical attention. Diagnostic laboratory findings are decreased blood pH (< 7.35), increased plasma lactate levels (> 5 mmol/L) and an increased anion gap and lactate/pyruvate ratio.
Renal function
GFR should be assessed before treatment initiation and regularly thereafter (see section 4.2). Sitagliptin Phosphate & Metformin Hydrochloride Tablets is contraindicated in patients with GFR < 30 mL/min and should be temporarily discontinued during conditions with the potential to alter renal function.
Hypoglycaemia
Patients receiving Sitagliptin Phosphate & Metformin Hydrochloride Tablets in combination with a sulphonylurea or with insulin may be at risk for hypoglycaemia. Therefore, a reduction in the dose of the sulphonylurea or insulin may be necessary.
Hypersensitivity reactions
Post-marketing reports of serious hypersensitivity reactions in patients treated with sitagliptin have been reported. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Onset of these reactions occurred within the first 3 months after initiation of treatment with sitagliptin, with some reports occurring after the first dose. If a hypersensitivity reaction is suspected, Sitagliptin Phosphate & Metformin Hydrochloride Tablets should be discontinued, other potential causes of the event should be assessed, and alternative treatment for diabetes should be instituted.
Bullous pemphigoid
There have been post-marketing reports of bullous pemphigoid in patients taking DPP-4 inhibitors including sitagliptin. If bullous pemphigoid is suspected, Sitagliptin Phosphate & Metformin Hydrochloride Tablets should be discontinued.
Surgery
Sitagliptin Phosphate & Metformin Hydrochloride Tablets must be discontinued at the time of surgery under general, spinal or epidural anaesthesia. Therapy may be restarted no earlier than 48 hours following surgery or resumption of oral nutrition and provided that renal function has been re-evaluated and found to be stable.
Administration of iodinated contrast agent
Intravascular administration of iodinated contrast agents may lead to contrast-induced nephropathy, resulting in metformin accumulation and an increased risk of lactic acidosis. Sitagliptin Phosphate & Metformin Hydrochloride Tablets should be discontinued prior to or at the time of the imaging procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Change in clinical status of patients with previously controlled type 2 diabetes
A patient with type 2 diabetes previously well controlled on Sitagliptin Phosphate & Metformin Hydrochloride Tablets who develops laboratory abnormalities or clinical illness (especially vague and poorly defined illness) should be evaluated promptly for evidence of ketoacidosis or lactic acidosis. Evaluation should include serum electrolytes and ketones, blood glucose and, if indicated, blood pH, lactate, pyruvate, and metformin levels. If acidosis of either form occurs, treatment must be stopped immediately and other appropriate corrective measures initiated.
DRUG INTERACTIONS
Carbonic Anhydrase Inhibitors
Clinical Impact: Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis.Concomitant use of these drugs with Sitagliptin Phosphate & Metformin Hydrochloride Tablets may increase the risk for lactic acidosis.
Intervention: Consider more frequent monitoring of these patients. Examples: Topiramate, zonisamide, acetazolamide or dichlorphenamide.
Drugs that Reduce Metformin Clearance
Clinical Impact: Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g.,organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis Intervention: Consider the benefits and risks of concomitant use with Sitagliptin Phosphate & Metformin Hydrochloride Tablets.
Examples: Ranolazine, vandetanib, dolutegravir, and cimetidine.
Alcohol
Clinical Impact: Alcohol is known to potentiate the effect of metformin on lactate metabolism. Intervention: Warn patients against alcohol intake while receiving Sitagliptin Phosphate & Metformin Hydrochloride Tablets.
Insulin Secretagogues or Insulin
Clinical Impact: Coadministration of Sitagliptin Phosphate & Metformin Hydrochloride Tablets with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia.
Intervention: Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin.
Drugs Affecting Glycemic Control Clinical Impact: Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control.
Intervention: When such drugs are administered to a patient receiving Sitagliptin Phosphate & Metformin Hydrochloride Tablets, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving Sitagliptin Phosphate & Metformin Hydrochloride Tablets, observe the patient closely for hypoglycemia. Examples: Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid.
FERTILITY, PREGNANCY AND LACTATION
Pregnancy
There are no adequate data from the use of sitagliptin in pregnant women. Studies in animals have shown reproductive toxicity at high doses of sitagliptin.
A limited amount of data suggests the use of metformin in pregnant women is not associated with an increased risk of congenital malformations. Animal studies with metformin do not indicate harmful effects with respect to pregnancy, embryonic or foetal development, parturition or postnatal development.
Sitagliptin Phosphate & Metformin Hydrochloride Tablets should not be used during pregnancy. If a patient wish to become pregnant or if a pregnancy occurs, treatment should be discontinued and the patient switched to insulin treatment as soon as possible.
Breast-feeding
No studies in lactating animals have been conducted with the combined active substances of this medicinal product. In studies performed with the individual active substances, both sitagliptin and metformin are excreted in the milk of lactating rats. Metformin is excreted in human milk in small amounts. It is not known whether sitagliptin is excreted in human milk. Sitagliptin Phosphate & Metformin Hydrochloride Tablets must therefore not be used in women who are breast-feeding.
Fertility
Animal data do not suggest an effect of treatment with sitagliptin on male and female fertility. Human data are lacking.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Sitagliptin Phosphate & Metformin Hydrochloride Tablets has no or negligible influence on the ability to drive and use machines. However, when driving or using machines, it should be taken into account that dizziness and somnolence have been reported with sitagliptin.
In addition, patients should be alerted to the risk of hypoglycaemia when Sitagliptin Phosphate & Metformin Hydrochloride Tablets is used in combination with a sulphonylurea or with insulin.
UNDESIRABLE EFFECTS
Dipeptidyl-peptidase-4 (DPP-4) inhibitors induced arthralgia: Dipeptidyl-peptidase-4 (DPP-4) inhibitors like sitagliptin, vildagliptin, Saxagliptin, etc induced arthralgia.
Summary of the safety profile
There have been no therapeutic clinical trials conducted with Sitagliptin Phosphate & Metformin Hydrochloride Tablets tablets however bioequivalence of Sitagliptin Phosphate & Metformin Hydrochloride Tablets with co-administered sitagliptin and metformin has been demonstrated. Serious adverse reactions including pancreatitis and hypersensitivity reactions have been reported. Hypoglycaemia has been reported in combination with sulphonylurea (13.8 %) and insulin (10.9 %).
Sitagliptin and metformin
Tabulated list of adverse reactions Adverse reactions are listed below as MedDRA preferred term by system organ class and absolute frequency (Table 1). Frequencies are defined as: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000) and not known (cannot be estimated from the available data).
Table 1: The frequency of adverse reactions identified from placebo-controlled clinical studies of sitagliptin and metformin alone, and post-marketing experience
Adverse reaction | Frequency of adverse reaction |
Blood and lymphatic system disorders |
|
thrombocytopenia | Rare |
Immune system disorders |
|
hypersensitivity reactions including anaphylactic responses*,† | Frequency not known |
Metabolism and nutrition disorders |
|
hypoglycaemia† | Common |
Nervous system disorders |
|
somnolence | Uncommon |
Respiratory, thoracic and mediastinal disorders |
|
interstitial lung disease* | Frequency not known |
Gastrointestinal disorders |
|
diarrhoea | Uncommon |
nausea | Common |
flatulence | Common |
constipation | Uncommon |
upper abdominal pain | Uncommon |
vomiting | Common |
acute pancreatitis*,†,‡ | Frequency not known |
fatal and non-fatal haemorrhagic and necrotizing | Frequency not known |
pancreatitis*,† |
|
Skin and subcutaneous tissue disorders |
|
pruritus* | Uncommon |
angioedema*,† | Frequency not known |
rash*,† | Frequency not known |
urticaria*,† | Frequency not known |
cutaneous vasculitis*,† | Frequency not known |
exfoliative skin conditions including Stevens-Johnson syndrome*,† – | Frequency not known |
bullous pemphigoid* | Frequency not known |
Musculoskeletal and connective tissue disorders |
|
arthralgia* | Frequency not known |
myalgia* | Frequency not known |
pain in extremity* | Frequency not known |
back pain* | Frequency not known |
arthropathy* | Frequency not known |
Renal and urinary disorders |
|
impaired renal function* | Frequency not known |
acute renal failure* | Frequency not known |
*Adverse reactions were identified through post-marketing surveillance.
‡See TECOS Cardiovascular Safety Study below.
Description of selected adverse reactions
Some adverse reactions were observed more frequently in studies of combination use of sitagliptin and metformin with other anti-diabetic medicinal products than in studies of sitagliptin and metformin alone. These included hypoglycaemia (frequency very common with sulphonylurea or insulin), constipation (common with sulphonylurea), peripheral oedema (common with pioglitazone), and headache and dry mouth (uncommon with insulin).
Sitagliptin
In monotherapy studies of sitagliptin 100 mg once daily alone compared to placebo, adverse reactions reported were headache, hypoglycaemia, constipation, and dizziness.
Among these patients, adverse events reported regardless of causal relationship to medicinal product occurring in at least 5 % included upper respiratory tract infection and nasopharyngitis. In addition, osteoarthritis and pain in extremity were reported with frequency uncommon (> 0.5 % higher among sitagliptin users than that in the control group).
Metformin
Gastrointestinal symptoms were reported very commonly in clinical studies and post-marketing use of metformin. Gastrointestinal symptoms such as nausea, vomiting, diarrhoea, abdominal pain and loss of appetite occur most frequently during initiation of therapy and resolve spontaneously in most cases. Additional adverse reactions associated with metformin include metallic taste (common); lactic acidosis, liver function disorders, hepatitis, urticaria, erythema, and pruritus (very rare). Long-term treatment with metformin has been associated with a decrease in vitamin B12 absorption which may very rarely result in clinically significant vitamin B12 deficiency (e.g., megaloblastic anaemia). Frequency categories are based on information available from metformin Summary of Product Characteristics available in the EU.
Paediatric population In clinical trials with Sitagliptin Phosphate & Metformin Hydrochloride Tablets in paediatric patients with type 2 diabetes mellitus aged 10 to 17 years, the profile of adverse reactions was generally comparable to that observed in adults. In paediatric patients on or not on background insulin, sitagliptin was associated with an increased risk of hypoglycaemia.
TECOS Cardiovascular Safety Study The Trial Evaluating Cardiovascular Outcomes with Sitagliptin (TECOS) included 7,332 patients treated with sitagliptin, 100 mg daily (or 50 mg daily if the baseline eGFR was ≥ 30 and < 50 mL/min/1.73 m2), and 7,339 patients treated with placebo in the intention-to-treat population. Both treatments were added to usual care targeting regional standards for HbA and CV risk factors. The overall incidence of serious adverse events in patients receiving sitagliptin was similar to that in patients receiving placebo. In the intention-to-treat population, among patients who were using insulin and/or a sulfonylurea at baseline, the incidence of severe hypoglycaemia was 2.7 % in sitagliptin-treated patients and 2.5 % in placebo-treated patients; among patients who were not using insulin and/or a sulfonylurea at baseline, the incidence of severe hypoglycaemia was 1.0 % in sitagliptin- treated patients and 0.7 % in placebo-treated patients. The incidence of adjudication-confirmed pancreatitis events was 0.3 % in sitagliptin-treated patients and 0.2 % in placebo-treated patients.
Reporting of side effects or suspected adverse reactions: If you experience any side effects, talk to your doctor or If you experience any side effects, talk to your doctor or pharmacist or report to indiadrugsafety@akums.in or report side effects directly by calling on the toll-free number 829 700 0060 or directly via the National Pharmacovigilance Program of India. By reporting side effects, you can help provide more information on the safety of this product.
OVERDOSE
During controlled clinical trials in healthy subjects, single doses of up to 800 mg sitagliptin were administered. Minimal increases in QTc, not considered to be clinically relevant, were observed in one study at a dose of 800 mg sitagliptin. There is no experience with doses above 800 mg in clinical studies. In Phase I multiple-dose studies, there were no dose-related clinical adverse reactions observed with sitagliptin with doses of up to 600 mg per day for periods of up to 10 days and 400 mg per day for periods of up to 28 days.
A large overdose of metformin (or co-existing risks of lactic acidosis) may lead to lactic acidosis which is a medical emergency and must be treated in hospital. The most effective method to remove lactate and metformin is haemodialysis.
In clinical studies, approximately 13.5 % of the dose was removed over a 3- to 4-hour haemodialysis session. Prolonged haemodialysis may be considered if clinically appropriate. It is not known if sitagliptin is dialysable by peritoneal dialysis.
In the event of an overdose, it is reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring (including obtaining an electrocardiogram), and institute supportive therapy if required.
PHARMACOLOGICAL PROPERTIES
Pharmacodynamic properties Pharmacotherapeutic group: Drugs used in diabetes, Combinations of oral blood glucose lowering drugs, ATC code: A10BD07
Sitagliptin
Mechanism of action Sitagliptin phosphate is an orally-active, potent, and highly selective inhibitor of the dipeptidyl peptidase 4 (DPP-4) enzyme for the treatment of type 2 diabetes. The DPP-4 inhibitors are a class of agents that act as incretin enhancers. By inhibiting the DPP-4 enzyme, sitagliptin increases the levels of two known active incretin hormones, glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). The incretins are part of an endogenous system involved in the physiologic regulation of glucose homeostasis. When blood glucose concentrations are normal or elevated, GLP-1 and GIP increase insulin synthesis and release from pancreatic beta cells. GLP-1 also lowers glucagon secretion from pancreatic alpha cells, leading to reduced hepatic glucose production. When blood glucose levels are low, insulin release is not enhanced and glucagon secretion is not suppressed. Sitagliptin is a potent and highly selective inhibitor of the enzyme DPP-4 and does not inhibit the closely-related enzymes DPP-8 or DPP-9 at therapeutic concentrations. Sitagliptin differs in chemical structure and pharmacological action from GLP-1 analogues, insulin, sulphonylureas or meglitinides, biguanides, peroxisome proliferator-activated receptor gamma (PPARγ) agonists, alpha-glucosidase inhibitors, and amylin analogues.
Metformin
Mechanism of action Metformin is a biguanide with antihyperglycaemic effects, lowering both basal and postprandial plasma glucose. It does not stimulate insulin secretion and therefore does not produce hypoglycaemia.
Metformin may act via three mechanisms:
- by reduction of hepatic glucose production by inhibiting gluconeogenesis and glycogenolysis
- in muscle, by modestly increasing insulin sensitivity, improving peripheral glucose uptake and utilisation
- by delaying intestinal glucose
Metformin stimulates intracellular glycogen synthesis by acting on glycogen synthase. Metformin increases the transport capacity of specific types of membrane glucose transporters (GLUT-1 and GLUT-4).
Pharmacokinetic Properties
Sitagliptin
Absorption
Following oral administration of a 100-mg dose to healthy subjects, sitagliptin was rapidly absorbed, with peak plasma concentrations (median Tmax) occurring 1 to 4 hours post-dose, mean plasma AUC of sitagliptin was 8.52 μMhr, Cmax was 950 nM. The absolute bioavailability of sitagliptin is approximately 87 %. Since co-administration of a high-fat meal with sitagliptin had no effect on the pharmacokinetics, sitagliptin may be administered with or without food.
Plasma AUC of sitagliptin increased in a dose-proportional manner. Dose-proportionality was not established for Cmax and C24hr (Cmax increased in a greater than dose-proportional manner and C24hr increased in a less than dose-proportional manner).
Distribution
The mean volume of distribution at steady state following a single 100-mg intravenous dose of sitagliptin to healthy subjects is approximately 198 litres. The fraction of sitagliptin reversibly bound to plasma proteins is low (38 %).
Biotransformation
Sitagliptin is primarily eliminated unchanged in urine, and metabolism is a minor pathway. Approximately 79 % of sitagliptin is excreted unchanged in the urine. Following a [14C] sitagliptin oral dose, approximately 16 % of the radioactivity was excreted as metabolites of sitagliptin. Six metabolites were detected at trace levels and are not expected to contribute to the plasma DPP-4 inhibitory activity of sitagliptin. In vitro studies indicated that the primary enzyme responsible for the limited metabolism of sitagliptin was CYP3A4, with contribution from CYP2C8.
In vitro data showed that sitagliptin is not an inhibitor of CYP isoenzymes CYP3A4, 2C8, 2C9, 2D6, 1A2, 2C19 or 2B6, and is not an inducer of CYP3A4 and CYP1A2. Elimination
Following administration of an oral [14C] sitagliptin dose to healthy subjects, approximately 100 % of the administered radioactivity was eliminated in faeces (13 %) or urine (87 %) within
one week of dosing. The apparent terminal t½ following a 100-mg oral dose of sitagliptin was approximately 12.4 hours. Sitagliptin accumulates only minimally with multiple doses. The renal clearance was approximately 350 mL/min.
Metformin
Absorption
After an oral dose of metformin, Tmax is reached in 2.5 h. Absolute bioavailability of a 500 mg metformin tablet is approximately 50-60 % in healthy subjects. After an oral dose, the non- absorbed fraction recovered in faeces was 20-30 %.
After oral administration, metformin absorption is saturable and incomplete. It is assumed that the pharmacokinetics of metformin absorption is non-linear. At the usual metformin doses and dosing schedules, steady state plasma concentrations are reached within 24-48 h and are generally less than 1 µg/mL. In controlled clinical trials, maximum metformin plasma levels (Cmax) did not exceed 5 µg/mL, even at maximum doses.
Food decreases the extent and slightly delays the absorption of metformin. Following administration of a dose of 850 mg, a 40 % lower plasma peak concentration, a 25 % decrease in AUC and a 35 min prolongation of time to peak plasma concentration was observed. The clinical relevance of this decrease is unknown.
Distribution
Plasma protein binding is negligible. Metformin partitions into erythrocytes. The blood peak is lower than the plasma peak and appears at approximately the same time. The red blood cells most likely represent a secondary compartment of distribution. The mean Vd ranged between 63 – 276 L.
Biotransformation
Metformin is excreted unchanged in the urine. No metabolites have been identified in humans. Elimination
Renal clearance of metformin is > 400 mL/min, indicating that metformin is eliminated by glomerular filtration and tubular secretion. Following an oral dose, the apparent terminal elimination half-life is approximately 6.5 h. When renal function is impaired, renal clearance is decreased in proportion to that of creatinine and thus the elimination half-life is prolonged, leading to increased levels of metformin in plasma.
INCOMPATIBILITIES
None stated.
STORAGE AND HANDLING INSTRUCTIONS
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
Other Products

Teliglim-MV

Telipanta 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.