- Home
- Teliglim-MV
Teliglim-MV
Teliglim-MV Tablet
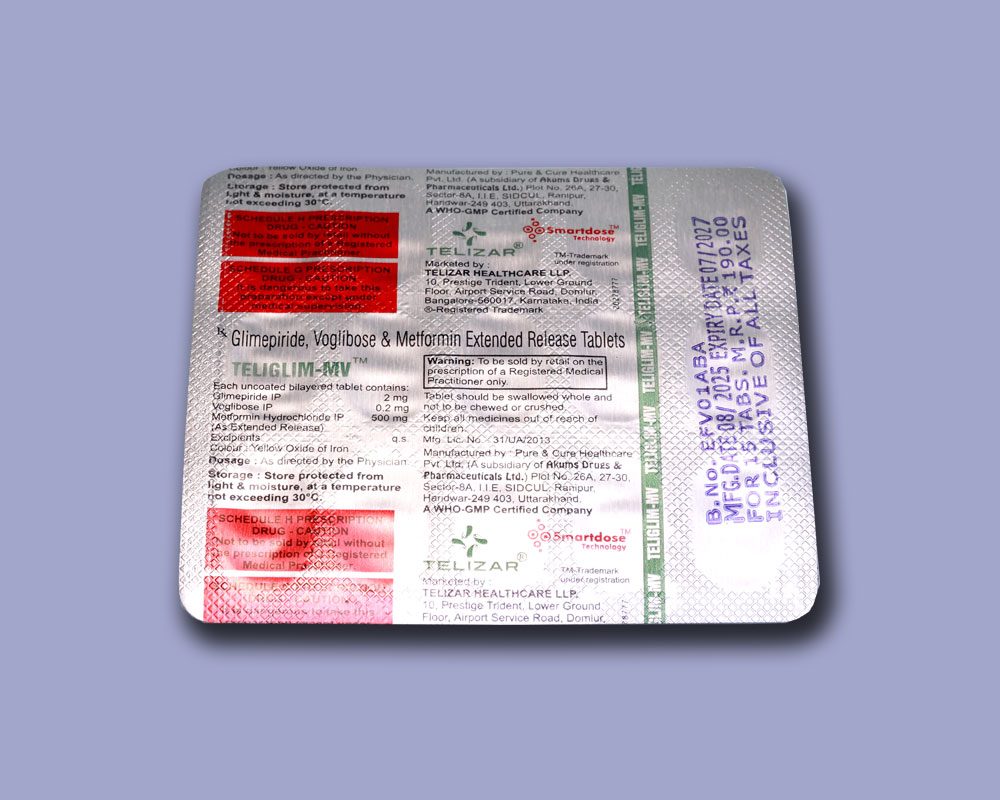
Each uncoated bilayered tablet contains:
Glimepiride IP 2 mg
Voglibose IP 0.2 mg
Metformin Hydrochloride IP 500 mg (As Extended Release)
Excipients q.s.
Colour : Yellow Oxide of Iron
PHARMACEUTICAL FORM
Uncoated bilayered tablet
THERAPEUTIC INDICATION
It is indicated in the treatment of patients with type II diabetes mellitus when diet, exercise and therapy with two agents do not result in adequate glycemic control.
DOSAGE AND ADMINISTRATION
General
The usual recommended dose for adults is 1 tablet once a day before meals or as directed by the Physician.
Special Populations:
Children: Data are insufficient to recommend pediatric use of this tablet.
Renal impairment: A GFR should be assessed before initiation of treatment with metformin containing products and at least annually thereafter. In patients at increased risk of further progression of renal impairment and in the elderly, renal function should be assessed more frequently, e.g. every 3-6 months.
The maximum daily dose of metformin should preferably be divided into 2-3 daily doses. Factors that may increase the risk of lactic acidosis should be reviewed before considering initiation of metformin in patients with GFR<60 mL/min.
Maximum Recommended Dose: The maximum recommended dose for glimepiride is 8 mg daily. The maximum recommended daily dose for metformin sustained-release is 2000 mg in adults. The daily dose should be titrated based on the glycemic control, in increments of 1 tablet only, corresponding to the lowest strength.
Additionally, voglibose tablets may be taken before the remaining meal, as prescribed by the physician. In order to avoid hypoglycemia the starting dose should not exceed the daily doses.
Method of administration: For oral administration only. Due to sustained release formulation, the tablets must be swallowed whole and not crushed or chewed.
CONTRAINDICATIONS
It is contraindicated in patients with known hypersensitivity or idiosyncrasy to any of its ingredients or to any other component of this product.
Glimepiride is contraindicated in followings:
- In patients hypersensitive to glimepiride, other sulfonylureas, other
- Pregnant and breast-feeding
- No experience has been gained concerning the use of glimepiride in patients with severe impairment of liver function and in dialysis In patients with severe impairment of hepatic function, change-over to insulin is indicated, not least to achieve optimal metabolic control.
- Voglibose is contraindicated in patients with:
- Severe ketosis or in a state of diabetic coma or
- Severe infections, before or after operation or with severe
- Gastrointestinal obstruction or predisposed to Metformin is contraindicated in followings:
- Any type of acute metabolic acidosis (such as lactic acidosis, diabetic ketoacidosis, diabetic pre-coma).
- Severe renal failure (GFR˂30ml/min).
- Acute conditions with the potential to alter renal function such as: Dehydration, severe infection, shock, and intravascular administration of iodinated contrast
- Acute or chronic disease which may cause tissue hypoxia such as: cardiac or respiratory failure, recent myocardial infarction, and
- Hepatic insufficiency.
- Acute alcohol intoxication, alcoholism.
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
This tablet should be administered carefully in patients who are receiving other antidiabetic drugs because hypoglycemia may occur.
Glimepiride
In exceptional stress situations (e.g. trauma, surgery, febrile infections) blood glucose regulation may deteriorate, and a temporary change to insulin may be necessary to maintain good metabolic control.
Hypoglycemia
All sulphonylurea drugs are capable of producing severe hypoglycemia. Proper patient selection, dosage, and instructions are important to avoid hypoglycemic episodes. When a patient stabilized on any diabetic regimen, is exposed to stress such as fever, trauma, infection, or surgery, a temporary loss of glycemic control may occur. At such times, it may be necessary to withhold the diabetic regime and temporarily administer insulin.
Cardiac effects
The administration of oral hypoglycemic drugs has been reported to be associated with increased cardiovascular mortality as compared to treatment with diet alone or diet plus insulin. In view of close similarities between the oral hypoglycemic drugs, this warning also applies for glimepiride.
In exceptional stress situations (e.g. trauma, surgery, febrile infections) blood glucose regulation may deteriorate, and a temporary change to insulin may be necessary to maintain good metabolic control.
In the initial weeks of treatment, the risk of hypoglycaemia may be increased and necessitates especially careful monitoring. Factors favoring hypoglycaemia include: Unwillingness or (more commonly in older patients) incapacity of the patient to cooperate; undernourishment, irregular mealtimes or skipped meals; imbalance between physical exertion and carbohydrate intake; alterations of diet, consumption of alcohol, especially in combination with skipped meals; impaired renal function, and severe impairment of liver function, and overdosage with glimepiride; certain uncompensated disorders of the endocrine system affecting carbohydrate metabolism or counter-regulation of hypoglycaemia (as for example in certain disorders of thyroid function and in anterior pituitary or corticoadrenal insufficiency) and concurrent administration of certain other medicines (see Interactions); Treatment with glimepiride in the absence of any indication.
If such risk factors for hypoglycaemia are present, it may be necessary to adjust the dosage of glimepiride or the entire therapy. This also applies whenever illness occurs during therapy or the patient’s life-style changes. Those symptoms of hypoglycaemia which reflect the body’s adrenergic counter regulation (see Adverse Reactions) may be milder or absent where hypoglycaemia develops gradually, in the elderly, and where there is autonomic neuropathy or where the patient is receiving concurrent treatment with beta- blockers, clonidine, reserpine, guanethidine or other sympatholytic drugs.
Hypoglycaemia can almost always be promptly controlled by immediate intake of carbohydrates (glucose or sugar). It is known from other sulfonylureas that, despite initially successful countermeasures, hypoglycaemia may recur. Patients must, therefore, remain under close observation.
Severe hypoglycaemia further requires immediate treatment and follow-up by a physician and, in some circumstances, in-patient hospital care.
Treatment of patients with G6PD-deficiency with sulfonylurea agents can lead to hemolytic anaemia. Since glimepiride belongs to the class of sulfonylurea agents, caution should be used in patients with G6PD-deficiency and a non-sulfonylurea alternative should be considered.
Voglibose
Voglibose should be administered with caution to the following patients: patients with history of laparotomy or ileus; patients with chronic intestinal disease accompanied by disturbance in digestion and absorption; patients with aggravating symptoms due to increased generation of intestinal gas (eg, Roemheld syndrome, severe hernia, and stenosis and ulcer of the large intestine) and patients with serious hepatic or renal disorders.
Other precautions:
All patients should continue their dietary restriction with a regular distribution of carbohydrate intake during the day. Overweight patients should continue their energy restricted diet.
The usual laboratory tests for diabetes monitoring should be performed regularly. Patients should be instructed and explained to recognize hypoglycemic symptoms and its management. When patients with diabetes are exposed to unusual stress such as fever, trauma, infection, or surgery, a temporary loss of control of blood glucose may occur. At such times insulin therapy may be necessary for some time.
Metformin
Lactic acidosis
Lactic acidosis is a rare, but serious, metabolic complication that can occur due to metformin accumulation during treatment with glimepiride and metformin combination therapy; when it occurs, it is fatal in approximately 50% of cases. When metformin is implicated as the cause of lactic acidosis, metformin plasma levels >5 μg/mL are generally found. Reported cases have occurred primarilyin diabetic patients with significant renal insufficiency and congestive heart failure.
Alcohol
Alcohol is known to potentiate the effect of metformin on lactate metabolism. Patients should be warned against excessive alcohol intake, while receiving metformin.
Cardiovascular
Cardiovascular collapse (shock) from whatever cause, acute congestive heart failure, acute myocardial infarction and other conditions characterized by hypoxemia have been associated with lactic acidosis and may also cause prerenal azotemia. When such events occur in patients on metformin therapy, the drug should be promptly discontinued.
In case of dehydration (severe diarrhoea or vomiting, fever or reduced fluid intake), metformin should be temporarily discontinued and contact with a health care professional is recommended.
Medicinal products that can acutely impair renal function (such as antihypertensives, diuretics and NSAIDs) should be initiated with caution in metformin-treated patients.
Other risk factors associated to lactic acidosis are excessive alcohol intake, hepatic insufficiency, inadequately controlled diabetes, ketosis, prolonged fasting and any conditions associated with hypoxia as well as concomitant use of medicinal products that may cause lactic acidosis.
Diagnosis: Patients and/or care-givers should be informed of the risk of lactic acidosis. In case of suspected symptoms, the patient should stop taking metformin and seek immediate medical attention Diagnostic laboratory findings are decreased blood pH (˂7.35), plasma lactate levels (˃5mmol/L), and an increased anion gap and lactate/pyruvate ratio.
Renal function
GFR should be assessed before treatment initiation and regularly thereafter. Metformin is contraindicated in patients with GFR<30 ml/min and should be temporarily discontinued in the presence of conditions that alter renal function, Decreased renal function in elderly subjects is frequent and asymptomatic. Special caution should be exercised in situations where renal function may become impaired, for example when initiating antihypertensive therapy or diuretic therapy and when starting therapy with an NSAID. Administration of iodinated contrast agent
Intravascular administration of iodinated contrast agents may lead to contrast induced nephropathy, resulting in metformin accumulation and an increased risk of lactic acidosis.
Metformin should be discontinued prior to, or at the time of the imaging procedure and not restarted until 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Surgery
Metformin must be discontinued at the time of surgery under general, spinal or epidural anaesthesia. Therapy may be restarted no earlier than 48 hours following surgery or resumption of oral nutrition and provided that renal function has been re-evaluated and found to be stable.
Regular monitoring of thyroid-stimulating hormone (TSH) levels is recommended in patients with hypothyroidism.
Long-term treatment with metformin has been associated with a decrease in vitamin B12 serum levels which may cause peripheral neuropathy. Monitoring of the vitamin B12 level is recommended.
Other precautions:
All patients should continue their diet with a regular distribution of carbohydrate intake during the day. Overweight patients should continue their energy-restricted diet.
The usual laboratory tests for diabetes monitoring should be performed regularly.
Metformin alone never causes hypoglycaemia, although caution is advised when it is used in combination with insulin or sulfonylureas.
DRUG INTERACTION
Glimepiride
CYP2C9 inducers (e.g. rifampicin) or inhibitors: Glimepiride is metabolized by cytochrome P450 2C9 (CYP2C9). Its metabolism is known to be influenced by concomitant administration of CYP2C9 inducers (e.g. rifampicin) or inhibitors (e.g. fluconazole). Results from an in-vivo interaction study reported in literature show that glimepiride AUC is increased approximately 2-fold by fluconazole, one of the most potent CYP2C9 inhibitors.
Based on the experience with glimepiride and with other sulfonylureas, the following interactions have to be mentioned. Potentiation of the blood-glucose-lowering effect and, thus, in some instances hypoglycaemia may occur when one of the following drugs is taken, for example: insulin and other oral antidiabetics; ACE inhibitors; anabolic steroids and male sex hormones; chloramphenicol; coumarin derivatives; cyclophosphamide; disopyramide; fenfluramine; fenyramidol; fibrates; fluoxetine; guanethidine; ifosfamide; MAO inhibitors; miconazole; fluconazole; paraaminosalicylic acid; pentoxifylline (high dose parenteral); phenylbutazone; azapropazone; oxyphenbutazone; probenecid; quinolones; salicylates; sulfinpyrazone; clarithromycin; sulfonamide antibiotics; tetracyclines; tritoqualine; trofosfamide.
Weakening of the blood-glucose-lowering effect and, thus raised blood glucose levels may occur when one of the following medicinal products is taken for example: acetazolamide; barbiturates; corticosteroids; diazoxide; diuretics; epinephrine (adrenaline) and other sympathomimetic agents; glucagon; laxatives (after protracted use); nicotinic acid (in high doses); oestrogens and progestogens; phenothiazines; phenytoin; rifampicin; thyroid hormones.
H2 receptor antagonists, beta-blockers, clonidine and reserpine may lead to either potentiation or weakening of the blood glucose- lowering effect.
Under the influence of sympatholytic drugs such as beta-blockers, clonidine, guanethidine and reserpine, the signs of adrenergic counter-regulation to hypoglycaemia may be reduced or absent. Both acute and chronic alcohol intake may potentiate or weaken the blood glucose-lowering action of glimepiride in an unpredictable fashion. The effect of coumarin derivatives may be potentiated or weakened.
Bile acid sequestrant: Colesevelam binds to glimepiride and reduces glimepiride absorption from the gastro-intestinal tract. No interaction was observed when glimepiride was taken at least 4 hours before colesevelam. Therefore glimepiride should be administered at least 4 hours prior to colesevelam.
Voglibose
When voglibose is used in combination with derivative(s) of sulfonylamide, sulfonylurea or biguanide, or with insulin, hypoglycemic symptoms may occur. Therefore, when used in combination with any of these drugs, care should be taken, such as starting the administration at a low dose.
When voglibose is administered concomitantly with drugs that enhance or diminish the hypoglycemic action of antidiabetic drugs, caution should be taken as this might additionally delay the action of voglibose on the absorption of carbohydrates. Examples of drugs enhancing the hypoglycemic action of antidiabetic drugs: alpha blockers, salicylic acid preparations, monoamine oxidase inhibitors and fibrate derivatives. Examples of drugs diminishing the hypoglycemic action of antidiabetic drugs: epinephrine, adrenocortical hormone, and thyroid hormone.
Voglibose does not affect the pharmacokinetics of warfarin; hence it can be safely administered along with warfarin.
Metformin
Concomitant use of Metformin not recommended:
Alcohol: Alcohol intoxication is associated with an increased risk of lactic acidosis, particularly in case of fasting or malnutrition or hepatic insufficiency.
Avoid consumption of alcohol and alcohol-containing medications.
Iodinated contrast agents: Metformin must be discontinued prior to, or at the time of the image procedure and not restarted until at least 48 hours after, provided that renal function has been re-evaluated and found to be stable.
Combinations requiring precautions for use:
Some medicinal products can adversely affect renal function which may increase the risk of lactic acidosis, e.g. NSAIDs, including selective cyclooxygenase (COX) II inhibitors, ACE inhibitors, angiotensin II receptor antagonists and diuretics, especially loop diuretics. When starting or using such products in combination with metformin, close monitoring of renal function is necessary.
Glucocorticoids (systemic and local routes), beta-2-agonists and diuretics have intrinsic hyperglycaemic activity. Inform the patient and perform more frequent blood glucose monitoring, especially at the beginning of treatment. If necessary, adjust the dosage of the antidiabetic drug during therapy with the other drug and upon its discontinuation. ACE-inhibitors may decrease the blood glucose levels. If necessary, adjust the dosage of the antidiabetic drug during therapy with the other drug and upon its discontinuation.
Metformin may decrease the anticoagulant effect of phenprocoumon. Therefore, a close monitoring is recommended. Levothyroxine can reduce the hypoglycemic effect of metformin. Monitoring of blood glucose levels is recommended, especially when thyroid hormone therapy is initiated or stopped, and the dosage of metformin must be adjusted if necessary
Organic cation transporters (OCT): Metformin is a substrate of both transporters OCT1 and OCT2. Co-administration of metformin with
Inhibitors of OCT1 (such as verapamil) may reduce efficacy of metformin. Inducers of OCT1 (such as rifampicin) may increase gastrointestinal absorption and efficacy of metformin. Inhibitors of OCT2 (such as cimetidine, dolutegravir, ranolazine, trimethoprime, vandetanib, isavuconazole) may decrease the renal elimination of metformin and thus lead to an increase in metformin plasma concentration.
Inhibitors of both OCT1 and OCT2 (such as crizotinib, olaparib) may alter efficacy and renal elimination of metformin.
Caution is therefore advised, especially in patients with renal impairment, when these drugs are coadministered with metformin, as metformin plasma concentration may increase. If needed, dose adjustment of metformin may be considered as OCT inhibitors/inducers may alter the efficacy of metformin.
USE IN SPECIFIC POPULATION
This tablet must not be taken during pregnancy. Otherwise there is risk of harm to the child. The patient must change over to insulin during pregnancy. To prevent possible ingestion with the breast milk and possible harm to the child, this tablet must not be taken by breast-feeding women. If necessary the patient must change over to insulin, or must stop breast-feeding.
Glimepiride
Pregnancy: Risk related to glimepiride, there are no adequate data from the use of glimepiride in pregnant women. Consequently, glimepiride should not be used during the whole pregnancy. In case of treatment by glimepiride, if the patient plans to become pregnant or if a pregnancy is discovered, the treatment should be switched as soon as possible to insulin therapy.
Lactation: The excretion in human milk is unknown. Glimepiride is excreted in rat milk. As other sulfonylureas are excreted in human milk and because there is a risk of hypoglycaemia in nursing infants, breast-feeding is advised against during treatment with glimepiride.
Voglibose
Pregnancy: The safety and effectiveness of voglibose in pregnant women has not been established. Animal studies have shown that voglibose is transferred to the foetus.
Lactation: Animal studies have shown a suppressive action of voglibose on body weight increase in newborns, mainly due to suppression of milk production resulting from inhibition of carbohydrate absorption in mother animals.
Metformin
Pregnancy: Uncontrolled diabetes during pregnancy (gestational or permanent) is associated with increased risk of congenital abnormalities and perinatal mortality. A limited amount of data from the use of metformin in pregnant women does not indicate an increased risk of congenital abnormalities. When the patient plans to become pregnant and during pregnancy, it is recommended that impaired glycaemic control or diabetes are not treated with metformin. For diabetes it is recommended that insulin should be used to maintain blood glucose levels as close to normal as possible to reduce the risk of malformations of the foetus.
Lactation: Metformin is excreted into human breast milk. No adverse effects were observed in breastfed newborns/infants. However, as only limited data are available, breastfeeding is not recommended during metformin treatment. A decision on whether to discontinue breast-feeding should be made, taking into account the benefit of breast-feeding and the potential risk to adverse effect on the child.
Fertility: Fertility of male or female rats was unaffected by metformin when administered at doses as high as 600mg/kg/day, which is approximately three times the maximum recommended human daily dose based on body surface area comparisons.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Alertness and reactions may be impaired due to hypo- or hyperglycemia, especially when beginning or after altering treatment or when this tablet is not taken regularly. This may, for example, affect the ability to drive or to operate machinery.
UNDESIRABLE EFFECTS
This tablet should be administered carefully in patients who are receiving other antidiabetic drugs because hypoglycemia may occur. The following CIOMS frequency rating is used, when applicable: Very common (≥10%); Common (≥1 and <10%); Uncommon (≥0.1 and <1%); Rare (≥0.01 and <0.1%); Very rare (<0.01%), Unknown (cannot be estimated from available data).
Glimepiride
Metabolism and nutrition disorders: As a result of the blood-glucose-lowering action of glimepiride, hypoglycaemia may occur, which may also be prolonged. Possible symptoms of hypoglycaemia include headache, ravenous hunger, nausea, vomiting, lassitude, sleepiness, disordered sleep, restlessness, aggressiveness, impaired concentration, impaired alertness and reactions, depression, confusion, speech disorders, aphasia, visual disorders, tremor, pareses, sensory disturbances, dizziness, helplessness, loss of self- control, delirium, cerebral convulsions, somnolence and loss of consciousness up to and including coma, shallow respiration and bradycardia.
In addition, signs of adrenergic counter-regulation may be present such as sweating, clammy skin, anxiety, tachycardia, hypertension, palpitations, angina pectoris, and cardiac arrhythmias. The clinical picture of a severe hypoglycaemic attack may resemble that of a stroke.
The symptoms nearly always subside when hypoglycaemia is corrected
Eye disorders: Especially at the start of treatment, there may be temporary visual impairment due to the change in blood glucose levels. The cause is a temporary alteration in the turgidity and hence the refractive index of the lens, this being dependent on the blood glucose level.
Gastrointestinal disorders: Occasionally, gastrointestinal symptoms such as nausea, vomiting, sensations of pressure or fullness in the epigastrium, abdominal pain and diarrhoea may occur. In isolated cases, there may be hepatitis, elevation of liver enzyme levels and/or cholestasis and jaundice, which may progress to life-threatening liver failure but can regress after withdrawal of glimepiride.
Dysgeusia (frequency not known).
Blood and lymphatic system disorders: Changes in the blood picture may occur: Rarely, thrombocytopenia and, in isolated cases, leucopenia, haemolytic anaemia, erythrocytopenia, granulocytopenia, agranulocytosis or pancytopenia may develop. Cases of severe thrombocytopenia with platelet count less than10,000/μl and thrombocytopenic purpura have been reported in post- marketing experience(frequency not known).
Skin and subcutaneous tissue disorders: Alopecia (frequency not known). General disorders: Occasionally, allergic or pseudoallergic reactions may occur, e.g. in the form of itching, urticarial or rashes. Such mild reactions may develop into serious reactions with dyspnoea and a fall in blood pressure, sometimes progressing to shock. In the event of urticaria a physician must therefore be notified immediately. In isolated cases, a decrease in serum sodium concentration and allergic vasculitis or hypersensitivity of the skin to light may occur.
Investigations: Glimepiride, like all sulfonylureas, can cause weight gain (frequency not known).
Voglibose
Gastrointestinal adverse effects such as diarrhoea, loose stools, abdominal pain, constipation, anorexia, nausea, vomiting, or heartburn may occur with the use of Voglibose. Also abdominal distention, increased flatus, and intestinal obstruction like symptoms due to an increase in intestinal gas, may occur with use of Voglibose.
When Voglibose is administered to patients with serious liver cirrhosis, hyperammonia may worsen with the development of constipation followed by disturbance of consciousness. Elevation of GOT (glutamate oxaloacetate), GPT (glutamatepyruvate transaminase), LDH (lactate dehydrogenase), alpha GPT (alpha glutamate pyruvate) or alkaline phosphatase may infrequently occur.
When Voglibose is used in combination with other antidiabetic drugs, hypoglycemia may occur (0.1% to <5%). Hypersensitivity: Rash and pruritus may rarely occur. In such a case, Voglibose tablets should be discontinued. Psychoneurologic: Headache may rarely occur.
Hematologic: Anemia; thrombocytopenia, and leucopenia may rarely occur.
Others: Numbness, edema of face, blurred vision, hot flushes, malaise, weakness, hyperkalemia, increased serum amylase, decreased HDL cholesterol, diaphoresis or alopecia, and perspiration.
Metformin
Gastrointestinal symptoms such as nausea, vomiting, diarrhea, abdominal pain and loss of appetite (>10%) are very common: these occur most frequently during initiation of therapy and resolve spontaneously in most cases. To prevent these gastrointestinal symptoms, it is recommended that metformin be taken in 2 or 3 daily doses during or after meals. A slow increase of the dose may also improve gastrointestinal tolerability.
- Metallic taste (3%) is
- Mild erythema has been reported in some hypersensitive The incidence of such effects is regarded as very rare (<0.01%).
- A decrease of vitamin B12 absorption with decrease of serum levels has been observed inpatients treated long-term with metformin and appears generally to be without clinical significance (<0.01%). However, cases of peripheral neuropathy in patients with vitamin B12 deficiency have been reported in post-marketing experience (frequency not known).
- Lactic acidosis (0.03 cases/1000 patient-years) is very rare.
- Hemolytic anemia (frequency unknown)
- Reduction of thyrotropin level in patients with hypothyroidism (frequency unknown)
- Hypomagnesemia in the context of diarrhea (frequency unknown)
- Encephalopathy (frequency unknown)
- Photosensitivity (frequency unknown)
- Hepatobiliary disorders: Reports of liver function tests abnormalities and hepatitis resolving upon metformin discontinuation. Reporting of side effects or suspected adverse reaction: If you experience any side effects, talk to your doctor or pharmacist or report to indiadrugsafety@akums.in. You can also report side effects directly via the National Pharmacovigilance Program of India by calling on 1800 180 By reporting side effects, you can help provide more information on the safety of this product.
OVERDOSE
Glimepiride
Signs and Symptoms: Acute overdosage as well as long-term treatment with too high a dose of glimepiride may lead to severe life- threatening hypoglycaemia.
Management: As soon as an overdose of glimepiride has been discovered, a physician must be notified without delay. The patient must immediately take sugar, if possible in the form of glucose, unless a physician has already undertaken responsibility for treating the overdose.
Careful monitoring is essential until the physician is confident that the patient is out of danger. It must be remembered that hypoglycaemia may recur after initial recovery. Patients who have ingested life-threatening amounts of glimepiride require detoxification (e.g. by gastric lavage and medicinal charcoal).
Voglibose
Voglibose competitively and reversibly inhibits the alpha glucosidase enzymes (glucoamylase, sucrase, maltase and isomaltase) in the brush border in the small intestine, which delays the hydrolysis of complex carbohydrates. It is unlikely to produce hypoglycemia in overdose, but abdominal discomfort and diarrhea may occur.
Metformin
Hypoglycaemia has not been seen with metformin doses of up to 85 g, although lactic acidosis has occurred in such circumstances. High overdose or concomitant risks of metformin may lead to lactic acidosis. Lactic acidosis is a medical emergency and must be treated in hospital. The most effective method to remove lactate and metformin is haemodialysis. Pancreatitis may occur in the context of a metformin overdose.
PHARMACOLOGICAL PROPERTIES
Pharmacodynamic properties
Glimepiride
Glimepiride is an orally active hypoglycaemic substance belonging to the sulphonylurea group. It may be used in non-insulin dependent (type 2) diabetes mellitus. Glimepiride acts mainly by stimulating insulin release from pancreatic beta cells. As with other sulfonylureas this effect is based on an increase of responsiveness of the pancreatic beta cells to the physiological glucose stimulus. In addition, glimepiride seems to have pronounced extra pancreatic effects also postulated for other sulfonylureas.
Voglibose
Voglibose is an alpha glucosidase inhibitor which inhibits the activity of alpha glucosidases that catalyse the decomposition of disaccharides into monosaccharides in the intestine, thereby delaying the digestion and absorption of carbohydrates, resulting in improvement of postprandial hyperglycaemia.
Metformin
Metformin is a biguanide oral anti-hyperglycaemic agent. Its mode of action is thought to be increased peripheral glucose utilization mediated by increased insulin sensitivity and inhibition of increased hepatic and renal gluconeogenesis. Metformin improves glucose tolerance in patients with type-2 diabetes (NIDDM), lowering both basal and postprandial plasma glucose. Metformin decreases hepatic glucose production, decreases intestinal absorption of glucose, and improves insulin sensitivity by increasing peripheral glucose uptake and utilization.
Pharmacokinetic properties
Absorption
The bioavailability of glimepiride after oral administration is complete. Food intake has no relevant influence on absorption, only the absorption rate is slightly diminished.
Voglibose is poorly absorbed after oral doses. Plasma concentrations after oral doses have usually been undetectable.
Metformin sustained-release: The absolute bioavailability of a metformin 500 mg tablet given under fasting conditions is approximately 50-60%. Following a single oral dose of metformin sustained-release, Cmax is achieved within 4-8 hours. Peak plasma levels are approximately 20% lower compared to the same dose of metformin immediate release. Both high and low fat meals had the same effect on the pharmacokinetics of sustained release.
Distribution
Glimepiride has a very low distribution volume (approx. 8.8 litres), which is roughly equal to the albumin distribution space, high protein binding (>99%).
After an 80 mg dose of voglibose (substantially higher than recommended dose), peak plasma levels of about 20 ng/mL were observed in 1-1.5 hours.
Metformin sustained-release: Metformin is negligibly bound to plasma proteins, in contrast to sulphonylureas, which are more than 90% protein bound. Metformin partitions into erythrocytes, most likely as a function of time.
Metabolism
Glimepiride is completely metabolized by oxidative biotransformation. The major metabolites are the cyclohexyl hydroxy methyl derivative (M1) and the carboxyl derivative (M2). Cytochrome P450 II C9 has been shown to be involved in the biotransformation of glimepiride to M1.
Voglibose is metabolized negligibly and rapidly excreted.
Metformin sustained-release: Metabolism studies with metformin sustained-release have not been conducted. However, intravenous single-dose studies in normal subjects demonstrate that metformin immediate release does not undergo hepatic metabolism or biliary excretion.
Excretion
Glimepiride: When 14C-glimepiride was given orally, approximately 60% of the total radioactivity was recovered in the urine in 7 days and M1 (predominant) and M2 accounted for 80-90% of that recovered in the urine.
When voglibose tablets were repeatedly administered to healthy male adults in a single dose of 0.2 mg, three times a day, for 7 consecutive days, voglibose was not detected in plasma or urine. Also, on administration of voglibose to 10 healthy male subjects in a single dose of 2 mg, voglibose was not detected in plasma or urine.
Metformin sustained-release: Following oral administration, approximately 90% of the absorbed drug is eliminated via the renal route within the first 24 hours, with a plasma elimination half-life of approximately 6.2 hours. In blood, the elimination half-life is approximately 17.6 hours, suggesting that the erythrocyte mass may be a compartment of distribution.
INCOMPATIBILITY
Not applicable.
SHELF LIFE
Refer on carton
STORAGE INSTRUCTIONS
Store protected from light & moisture, at a temperature not exceeding 30oC.
Keep all medicines out of reach of children.
Tablet should be swallowed whole and not chewed or crushed.
Other Products

Tezapride M1

Tezapride M2
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.