- Home
- Dipitel 5
Dipitel 5
Dipitel 5 Tablets
Dipitel 5 is an amlodipine besylate IP tablet containing 5 mg amlodipine, used for managing hypertension and improving cardiovascular health. This uncoated tablet helps relax blood vessels, ensuring smooth blood flow and effective long-term blood pressure control.
COMPOSITION
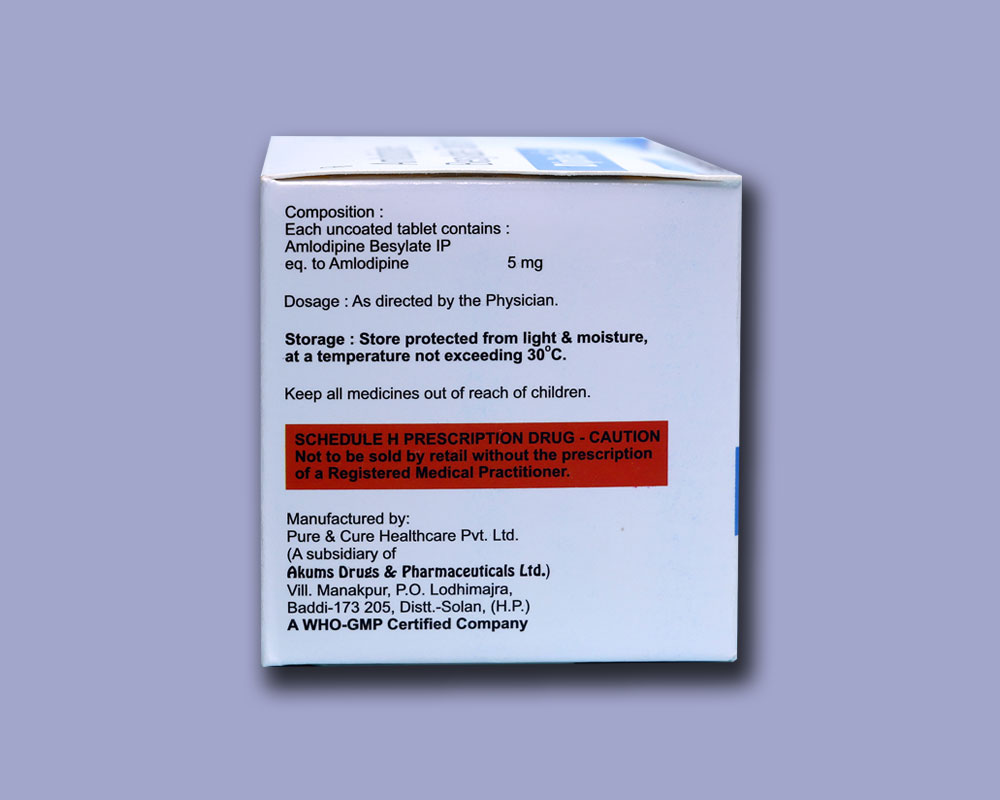
Each uncoated tablet contains :
Amlodipine Besylate IP
eq. to Amlodipine 5 mg
PHARMACEUTICAL FORM
Uncoated Tablet
THERAPEUTIC INDICATION
Amlodipine tablet is indicated:
- In treatment of hypertension and angina pectoris.
- To reduce of fatal coronary heart disease and non-fatal myocardial infarction, and to reduce the risk of stroke.
- To reduce the risk of coronary revascularization procedures and the need for hospitalization due to angina in patients
with coronary artery.
DOSAGE AND ADMINISTRATION
Posology
The recommended dosage is 1 tablet once daily or as directed by the Physician.
Adults
For both hypertension and angina the usual initial dose is 5 mg amlodipine once daily which may be increased to a maximum dose of 10 mg depending on the individual patient’s response. For angina, Amlodipine may be used as monotherapy or in combination with other antianginal medicinal products in patients with angina that is refractory to nitrates and/or to adequate doses of beta blockers. In hypertensive patients, Amlodipine has been used in combination with a thiazide diuretic, Alpha blocker, beta blockers,or an angiotensin-converting enzyme inhibitors, No dose adjustment of Amlodipine is required upon concomitantadministration of thiazide diuretics, beta blockers, and angiotensin-converting enzyme inhibitors.
Special populations Paediatric population: Children with hypertension from 6 years to 17 years of age. The recommended antihypertensive oral dose in pediatric patients ages 6-17 years is 2.5 mg once daily as a starting dose, up-titrated to 5 mg once daily if blood pressure goal is not achieved after 4 weeks. Doses in excess of 5 mg daily have not been studied in pediatric patients. The 2.5 mg dose cannot be obtained with Amlodipine tablets 5 mg and 10mg as these tablets are not manufactured to break into two equal halves.
Children under 6 years old: The effect of amlodipine on blood pressure in patients less than 6 years of age is not known.
Use in the elderly: Amlodipine, used at similar doses in elderly or younger patients, is equally well tolerated. Therefore normal dosage regimens are recommended, but increase of the dosage should take place with care.
Patients with hepatic impairment: Dosage recommendations have not been established in patients with mild to moderate hepatic impairment; therefore dose selection should be cautious and should start at the lower end of the dosing range. The pharmacokinetics of amlodipine have not been studied in severe hepatic impairment. Amlodipine should be initiated at the lowest dose and titrated slowly in patients with severe hepatic impairment.
Patients with renal impairment: Changes in amlodipine plasma concentrations are not correlated with degree of renal impairment, therefore the normal dosage is recommended. Amlodipine is not dialysable.
Method of administration: For oral use only.
CONTRAINDICATIONS
Amlodipine is contraindicated in patients with:
Hypersensitivity to dihydropyridines derivatives, amlodipine or to any of the excipients.
Shock (including cardiogenic shock), obstruction of the outflow tract of the left ventricle (e.g., high grade aortic stenosis),unstable angina (excluding Prinzmetal’s angina), severe hypotension, haemodynamically unstable heart failure afteracute myocardial. infarction.
SPECIAL WARNINGS AND PRECAUTIONS FOR USE
The safety and efficacy of amlodipine in hypertensive crisis has not been established.
Use in patients with heart failure: Patients with heart failure should be treated with caution. Calcium channel blockers, including amlodipine, should be used with caution in patients with congestive heart failure, as they may increase the risk of future cardiovascular events and mortality.
Use in patients with impaired hepatic function: As with all calcium antagonists, amlodipine’s half life is prolonged and AUC values are higher in patients with impaired liver function and dosage recommendations have not been established. The drug should therefore be administered with caution in these patients. Amlodipine should therefore be initiated at the
lower end of the dosing range and caution should be used, both on initial treatment and when increasing the dose. Slow dose titration and careful monitoring may be required in patients with severe hepatic impairment.
Elderly patients: In the elderly increase of the dosage should take place with care.
Patients with renal impairment: Amlodipine may be used in such patients at normal doses. Changes in amlodipine plasma concentrations are not correlated with degree of renal impairment. Amlodipine is not dialysable. There are no data to support the use of amlodipine alone, during or within one month of a myocardial infarction.
DRUG INTERACTION
In vitro data from studies with human plasma, indicate that amlodipine has no effect on protein binding of digoxin, phenytoin, warfarin or indomethacin. Consumption of grapefruit/grapefruit juice should be avoided while taking amlodipine. The intake of grapefruit juice may result in increased plasma amlodipine concentrations, which may enhance the blood pressure lowering effects of amlodipine. This interaction has been observed with other dihydropyridine calcium antagonists and represents a class effect.
Effects of other medicinal products on amlodipine
CYP3A4 inhibitors: Concomitant use of amlodipine with strong or moderate CYP3A4 inhibitors (protease inhibitors, azole antifungals, macrolides like erythromycin or clarithromycin, verapamil or diltiazem) may give rise to significant increase in amlodipine exposure resulting in an increased risk of hypotension. The clinical translation of these PK variations may be more pronounced in the elderly. Clinical monitoring and dose adjustment may thus be required.
CYP3A4 inducers: Upon co-administration of known inducers of the CYP3A4, the plasma concentration of amlodipine may vary. Therefore, blood pressure should be monitored and dose regulation considered both during and after concomitant medication particularly with strong CYP3A4 inducers (e.g. rifampicin, hypericum perforatum).
Dantrolene (infusion): Due to risk of hyperkalemia, it is recommended that the co- administration of calcium channel blockers such as amlodipine be avoided in patients susceptible to malignant hyperthermia and in the management of malignant hyperthermia.
Effects of amlodipine on other medicinal products
The blood pressure lowering effects of amlodipine adds to the blood pressure – lowering effects of other medicinal products with antihypertensive properties.
Tacrolimus: There is a risk of increased tacrolimus blood levels when co-administered with the amlodipine. In order to avoid toxicity of tacrolimus, use of amlodipine in a patient treated with tacrolimus requires monitoring of tacrolimus blood levels and dose adjustment when appropriate.
Cyclosporine: Consideration should be given for monitoring cyclosporine levels in renal transplant patients on amlodipine, and cyclosporine dose reductions should be made as necessary.
Simvastatin: Co-administration of multiple doses of 10mg of amlodipine with 80mg simvastatin resulted in a 77% increase in exposure to simvastatin compared to simvastatin alone. Limit the dose of simvastatin in patients on amlodipine to 20mg daily.
Cimetidine: Co-administration of amlodipine with cimetidine did not alter the pharmacokinetics of amlodipine.
Sildenafil: When amlodipine and sildenafil were used in combination, each agent independently exerted its own blood pressure lowering effect.
Warfarin: In healthy male volunteers, the co-administration of amlodipine does not significantly alter the effect of warfarin on prothrombin response time. Co-administration of amlodipine with warfarin did not change the warfarin prothrombin response time.
In clinical interaction studies, amlodipine did not affect the pharmacokinetics of atorvastatin, digoxin or warfarin.
FERTILITY, PREGNANCY AND LACTATION
Pregnancy: The safety of amlodipine in human pregnancy has not been established. In animal studies, reproductive toxicity was observed at high doses.Use in pregnancy is only recommended when there is no safer alternative and when the disease itself carries greater risk for the mother and foetus.
Breast-feeding: Amlodipine is excreted in human milk. The proportion of the maternal dose received by the infant has been estimated with an interquartile range of 3-7%, with a maximum of 15%. The effect of amlodipine on infants is unknown. A decision on whether to continue/discontinue breast- feeding or to continue/discontinue therapy with amlodipine should be made taking into account the benefit of breast-feeding to the child and the benefit of amlodipine therapy to the mother.
Fertility: Reversible biochemical changes in the head of spermatozoa have been reported in some patients treated by calcium channel blockers. Clinical data are insufficient regarding the potential effect of amlodipine on fertility. In one rat study, adverse effects were found on male fertility.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Amlodipine can have minor or moderate influence on the ability to drive and use machines. If patients taking amlodipine suffer from dizziness, headache, fatigue or nausea the ability to react may be impaired. Caution is recommended especially at the start of treatment.
UNDESIRABLE EFFECTS / ADVERSE DRUG REACTION
Gingival Hypertrophy and Alopecia as an adverse drug reaction has been reported with amlodipine.
The most commonly reported adverse reactions during amlodipine treatment are somnolence, dizziness, headache, palpitations, flushing, abdominal pain, nausea, ankle swelling, oedema and fatigue. The following adverse reactions have been observed and reported during treatment with amlodipine with the following frequencies: Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1,000 to <1/100); rare (≥1/10,000 to <1/1,000); very rare (<1/10,000). Within each frequency grouping, adverse reactions are presented in the order of decreasing seriousness:
Blood and lymphatic system disorders: Very rare: Leukocytopenia, thrombocytopenia.
Immune system disorders: Very rare: Allergic reactions.
Metabolism and nutrition disorders: Very rare: hyperglycaemia.
Psychiatric disorders: Uncommon: Depression, mood changes (including anxiety), insomnia. Rare: confusion.
Nervous system disorders: common: Somnolence, dizziness, headache (especially at the beginning of the treatment).
Uncommon: Tremor, dysgeusia, syncope, hypoaesthesia, paraesthesia. Very rare: Hypertonia, peripheral neuropathy.
Ear and labyrinth disorders: Common: Visual disturbance (including diplopia). Uncommon: Tinnitus.
Cardiac and Vascular disorders: Common: Palpitations. Uncommon; arrhythmia (including bradycardia, ventricular
tachycardia and atrial fibrillation). very rare: Myocardial infarction.
Vascular disorders: Common: flushing. Uncommon: hypotension. Very rare: vasculitis.
Respiratory, thoracic and mediastinal disorders: Common: dyspnoea. Uncommon: cough, rhinitis.
Gastrointestinal disorders: Common: Abdominal pain, nausea, dyspepsia, altered bowel habits (including diarrhoea
and constipation). Uncommon: Vomiting, dry mouth. Very rare: Pancreatitis, gastritis, gingival hyperplasia.
Hepatobiliary disorders: Very rare: Hepatitis, jaundice, hepatic enzyme increased.
Skin and subcutaneous tissue disorders: Uncommon: Alopecia, purpura, skin discolouration, hyperhidrosis, pruritus,
rash, exanthema, urticaria. Very rare: Angioedema, erythema multiforme, exfoliative dermatitis, Stevens-Johnson
syndrome, Quincke oedema, photosensitivity. Unknown: Toxic Epidermal Necrolysis.
Musculoskeletal and connective tissue disorders: Common:Ankle swelling, muscle cramps. Uncommon: Arthralgia,
myalgia, back pain.
Renal and urinary disorders: Uncommon: Micturition disorder, nocturia, increased urinary frequency.
Reproductive system and breast disorders: Uncommon: Impotence, gynaecomastia.
General disorders and administration site conditions: Very common: Oedema. Common: Fatigue, asthenia.
Uncommon: Chest pain, pain, malaise.
Investigations: Uncommon: Weight increased, weight decreased.
Exceptional cases of extrapyramidal syndrome have been reported.
OVERDOSE
Overdosage of amlodipine could result in excessive peripheral vasodilatation and possibly reflex tachycardia. Marked and probably prolonged systemic hypotension up to and including shock with fatal outcome have been reported. Administration of activated charcoal to healthy volunteers immediately or up to two hours after ingestion of amlodipine
10mg has been shown to significantly decrease amlodipine absorption. Gastric lavage may be worthwhile in some cases. Clinically significant hypotension due to amlodipine overdosage calls for active cardiovascular support including frequent monitoring of cardiac and respiratory function, elevation of extremities, and attention to circulating fluid volume and urine
output. A vasoconstrictor may be helpful in restoring vascular tone and blood pressure, provided that there is no contraindication to its use. Intravenous calcium gluconate may be beneficial in reversing the effects of calcium channel blockade. Since amlodipine is highly protein-bound, dialysis is not likely to be of benefit.
PHARMACOLOGICAL PROPERTIES
Pharmacotherapeutic group: Calcium channel blockers – Dihydropyridine derivatives. Amlodipine is a calcium ion influx inhibitor of the dihydropyridine group (slow channel blocker or calcium ion antagonist) and inhibits the transmembrane influx of calcium ions into cardiac and vascular smooth muscle. The mechanism of the antihypertensive action of amlodipine is due to a direct relaxant effect on vascular smooth muscle. The precise mechanism by which amlodipine relieves angina has not been fully determined but amlodipine reduces total ischaemic burden by the following two actions: Amlodipine dilates peripheral arterioles and thus, reduces the total peripheral resistance (afterload) against which the heart works. Since the heart rate remains stable, this unloading of the heart reduces myocardial energy consumption and oxygen requirements. The mechanism of action of amlodipine also probably involves dilatation of the main coronary arteries and coronary arterioles, both in normal and ischaemic regions. This dilatation increases myocardial oxygen delivery in patients with coronary artery spasm (Prinzmetal’s or variant angina). In patients with hypertension, once daily dosing provides clinically significant reductions of blood pressure in both the supine and standing positions throughout the 24 hour interval. Due to the slow onset of action, acute hypotension is not a feature of amlodipine administration. In patients with angina, once daily administration of amlodipine increases total exercise time, time to angina onset, and time to 1mm ST segment depression, and decreases both angina attack frequency and glyceryl trinitrate tablet consumption. Amlodipine has not been associated with any adverse metabolic effects or changes in plasma lipids and is suitable for use in patients with asthma, diabetes, and gout.
PHARMACOKINETIC PROPERTIES
Absorption: After oral administration of therapeutic doses, amlodipine is well absorbed with peak blood levels between 6-12 hours post dose. Absolute bioavailability has been estimated to be between 64 and 80%. The bioavailability of amlodipine is not affected by food intake.
Distribution: The volume of distribution is approximately 21 l/kg. In vitro studies have shown that approximately 97.5% of circulating amlodipine is bound to plasma proteins.
Metabolism: Amlodipine is extensively metabolized by the liver to inactive metabolites with 10% of the parent compound.
Excretion: About 60% of amlodipine metabolites excreted in the urine. The terminal plasma elimination half-life is about 35-50 hours and is consistent with once daily dosing.
INCOMPATIBILITY
None stated.
STORAGE INSTRUCTIONS
Store protected from light & moisture, at a temperature not exceeding 30°C.
Keep all medicines out of reach of children.
Other Products
Telmitel 40

Cilnidtel T 40
Get in touch with us.
We’d love to hear from you.
Wish to know more about us?
We’re constantly working to develop innovative healthcare products
Providing real goodness to patients and making a difference to their lives is of prime importance for us at Telizar Healthcare.